NEET Organic Chemistry 2026 Chapter-wise Topics, Key Reactions and Preparation Strategy
Most students treat Organic Chemistry like a memory test. They write down reactions, read them twice and hope something sticks. By the time the mock test arrives, nothing does — because reactions learned without understanding the why behind them simply do not stay.
Here is the truth: Organic Chemistry in NEET 2026 is not a memory test. It is a pattern recognition test. The NTA uses the same 4–5 core reaction mechanisms — dressed in different functional groups — across every paper. Once you understand those mechanisms, a question you have never seen before suddenly becomes one you can solve.
This guide covers the 8 chapters that matter, the reactions that actually appear in NEET, the ones most students confuse, and a revision approach that makes sense of all of it. No bloated reaction lists. No chapters you do not need. Just what works.
How Much Organic Chemistry Appears in NEET — Chapter Weightage and Priority
Before getting into chapters, it helps to see exactly how much Organic Chemistry is worth — and where the marks are actually sitting.
Organic Chemistry contributes roughly 34% of the total Chemistry section in NEET — more than Inorganic and Physical Chemistry individually. That translates to approximately 13–15 questions and 52–60 marks out of the 180-mark Chemistry section.
The split across all three Chemistry sections looks like this:
| Chemistry Section | Avg. Questions | Approx. Marks | Weightage |
| Organic Chemistry | 13–15 | 52–60 | ~34% |
| Inorganic Chemistry | 12–14 | 48–56 | ~31% |
| Physical Chemistry | 12–14 | 48–56 | ~31% |
| Chemistry Total | 45 | 180 | 100% |
Three sections, nearly equal weight — but Organic is slightly ahead. This means if you score well in Organic Chemistry, you are not just covering one-third of Chemistry, you are building a buffer that protects your total score.
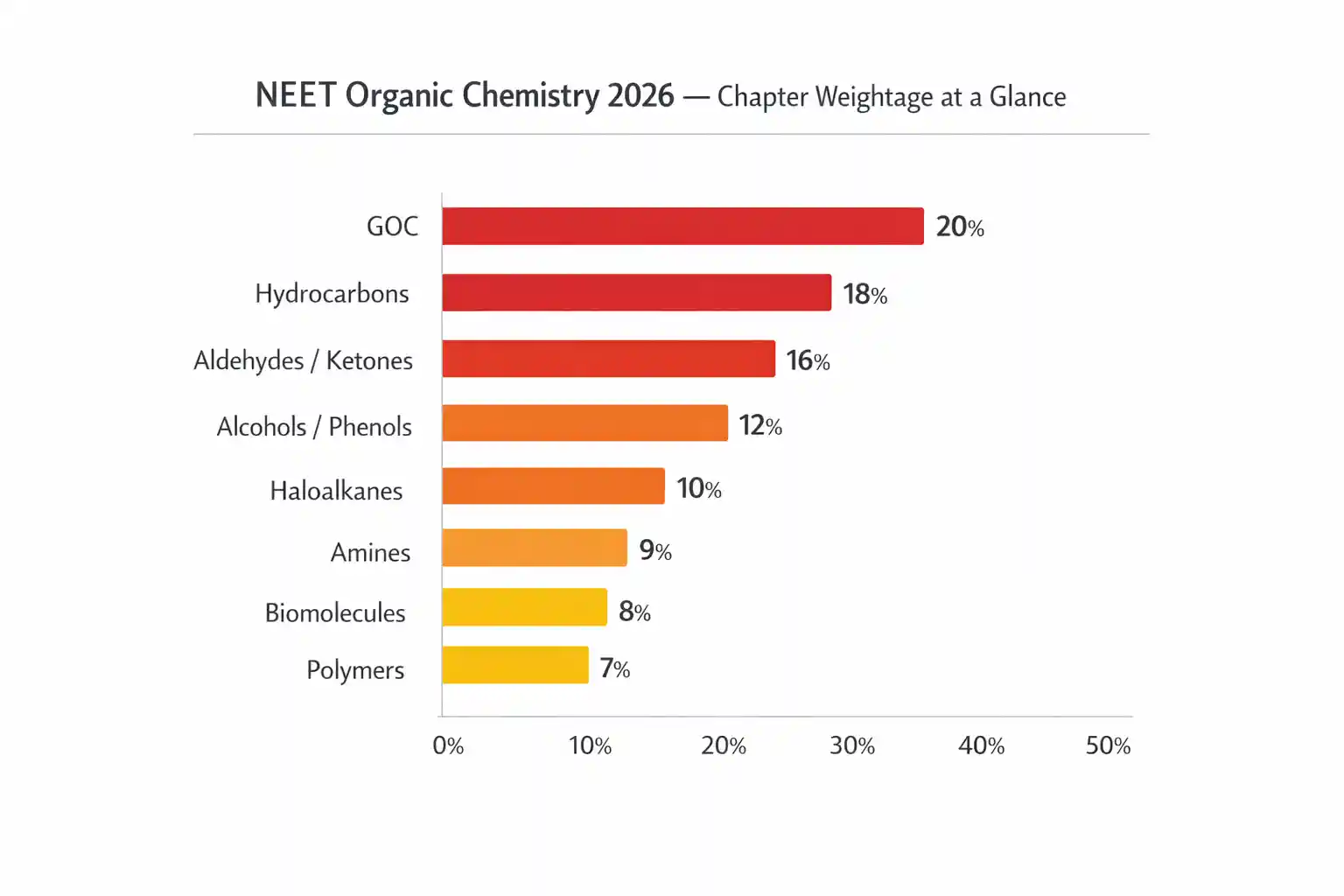
The 6 Chapters That Generate 90% of NEET Organic Chemistry Marks
Not all 8 Organic Chemistry chapters carry the same weight. Six chapters consistently dominate — the other two are secondary.
| Chapter | Class | Avg. Questions (Last 5 Years) | Weightage | Priority |
| Aldehydes, Ketones & Carboxylic Acids | 12 | 3–4 | ~9% | 🔴 Must-do first |
| General Organic Chemistry (GOC) | 11 | 2–3 | ~7% | 🔴 Must-do first |
| Hydrocarbons | 11 | 2–3 | ~6% | 🔴 Must-do |
| Alcohols, Phenols & Ethers | 12 | 2–3 | ~4–5% | 🟠 High priority |
| Haloalkanes & Haloarenes | 12 | 1–2 | ~4–5% | 🟠 High priority |
| Amines | 12 | 1–2 | ~4–5% | 🟠 High priority |
| Biomolecules | 12 | 1–2 | ~3% | 🟡 Moderate |
| Polymers & Chemistry in Everyday Life | 12 | 1 | ~2% | 🟡 Low — last |
Aldehydes, Ketones and Carboxylic Acids is the single highest-yield chapter in all of Organic Chemistry — it alone contributes 9% of the Chemistry section. If you have limited time, this chapter gets the most hours. Not because it is easy — but because the return on investment is the highest.
GOC (General Organic Chemistry) has to come first regardless of weightage. It is the foundation that every other chapter builds on — reactions in Haloalkanes, Alcohols, and Carbonyls will not make sense until you understand inductive effects, resonance and carbocation stability. This is the chapter most students either skip entirely or revise too lightly.
Organic Chemistry Marks — Where Students Gain and Where They Lose
Here is something most guides will not tell you: students who score 48+ marks in Organic Chemistry (out of ~60) are not smarter — they make fewer avoidable errors.
The gap between a 32-mark and a 52-mark Organic Chemistry performance is almost never about not knowing the content. It is about three patterns:
- Reagent confusion — two reagents that look similar (PCC vs K₂Cr₂O₇, NaBH₄ vs LiAlH₄) give different products, and students mix them up under pressure
- Mechanism applied to wrong substrate — SN1 and SN2 both involve substitution, but the conditions that trigger each are opposite, and the NTA regularly tests this distinction
- Named reactions misattributed — students know the Aldol condensation happens with aldehydes but cannot recall whether it requires a dilute base or concentrated acid
These are not knowledge gaps. They are precision gaps — and the solutions to all three are in Section 6 and Section 8 of this guide.
GOC — Why You Must Understand This Before Any Other Organic Chapter
Here is a question most students get wrong in their first mock: “Which carbocation is more stable — tertiary butyl or secondary propyl?”
Students who memorised the stability order answer it correctly. Students who understand why it is true can answer every variation of this question — including ones they have never seen. That difference is exactly what GOC teaches you.
GOC (General Organic Chemistry) is not a chapter you study to answer 2–3 GOC questions. It is the framework that makes every other Organic chapter make sense. Haloalkanes, Hydrocarbons, Aldehydes, Amines — every reaction in those chapters traces back to one or more concepts from GOC. Students who skip GOC or treat it lightly spend the next four chapters memorising reactions that they could be predicting.
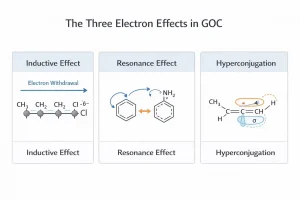
The Three Electron Effects That Predict Almost Every Organic Reaction
The NTA does not ask you to define these effects. It uses them in disguise — as questions about acidity, basicity, stability and reaction direction.
- Inductive Effect (−I and +I)
Electron-withdrawing groups (like −NO₂, −CN, −COOH, halogens) pull electrons toward themselves through sigma bonds — destabilising carbanions and stabilising carbocations nearby. Electron-donating groups (like −CH₃, −C₂H₅, alkyl groups) push electrons away — doing the opposite.
The NTA tests this as: “Which of the following acids is strongest?” followed by four options with different substituents. You do not need to memorise the answer — you need to know that more electron-withdrawing groups near the −COOH increase acidity by stabilising the carboxylate anion.
- Resonance Effect (+R and −R)
When electrons delocalise across a conjugated system — through pi bonds or lone pairs — the molecule becomes more stable and more predictable in its reactions.
This is why phenol is a stronger acid than ethanol (the phenoxide ion is resonance-stabilised), and why aniline is a weaker base than aliphatic amines (the lone pair on nitrogen is delocalised into the benzene ring, making it less available for protonation).
- Hyperconjugation
This is the effect students most often dismiss — and then lose marks over. Hyperconjugation explains why more substituted alkenes (and carbocations) are more stable. The more alpha-hydrogen atoms attached to the adjacent carbon, the greater the stabilisation through sigma-pi overlap.
The NTA tests hyperconjugation through carbocation stability, alkene stability, and sometimes directly through “which statement about hyperconjugation is correct” type questions.
Carbocation Stability — The One Concept Behind SN1, Rearrangements and Markovnikov’s Rule
If GOC is the foundation of Organic Chemistry, carbocation stability is the foundation of GOC.
The stability order is: Tertiary (3°) > Secondary (2°) > Primary (1°) > Methyl
This single sequence explains:
- Why SN1 reactions prefer tertiary substrates — they form stable tertiary carbocations as intermediates
- Why Markovnikov’s rule gives the product it does — the proton adds to the carbon that forms the more stable (more substituted) carbocation
- Why carbocation rearrangements happen — a less stable carbocation will rearrange via a hydride or methyl shift to become a more stable one
The NTA has tested all three of these applications in the last five years — sometimes directly, sometimes embedded in a multi-step reaction question.
One thing most students get wrong: they memorise that tertiary is most stable but cannot explain why. The reason is the combination of hyperconjugation (more alpha-H, more stabilisation) and the inductive effect (alkyl groups are +I, donating electron density to the positive carbon). Both effects increase as you move from primary to tertiary.
Once you understand the why, the order is impossible to forget — because you can reconstruct it from first principles.
Hydrocarbons — NEET-Relevant Reactions and What the NTA Actually Tests
Hydrocarbons contributes 2–3 questions per paper — drawn from three sub-chapters: Alkanes/Alkenes, Alkynes and Benzene. The NTA does not ask you to write full mechanisms in Hydrocarbons questions. It asks you to predict products — and you can do that reliably with the rules below.
The most common student mistake here: studying all hydrocarbon reactions from NCERT equally — including combustion of alkanes and Free Radical halogenation details — when the NTA draws almost entirely from just six specific reaction types.
Alkanes and Alkenes — Reactions, Addition Rules and NTA Question Types
What the NTA actually tests from Alkanes:
Almost nothing from alkane reactions except free radical halogenation order (3°H > 2°H > 1°H) and the fact that alkanes undergo substitution — not addition. One question every two to three papers, no more.
What the NTA consistently tests from Alkenes:
| Reaction | Reagent | Product | NTA Trap |
| Markovnikov addition | HBr (no peroxide) | H adds to less substituted C | Confused with anti-Markovnikov |
| Anti-Markovnikov addition | HBr + peroxide (ROOR) | H adds to more substituted C | Only HBr — not HCl or HI |
| Ozonolysis (reductive) | O₃ then Zn/H₂O | Aldehydes or ketones (no further oxidation) | Confused with oxidative ozonolysis |
| Ozonolysis (oxidative) | O₃ then H₂O₂ | Carboxylic acids from terminal alkenes | When to use which is the tested distinction |
| Hydroboration-oxidation | B₂H₆ then H₂O₂/OH⁻ | Anti-Markovnikov + syn addition | Stereochemistry aspect sometimes tested |
The peroxide effect is the most frequently confused rule in Hydrocarbons. Anti-Markovnikov addition only happens with HBr in the presence of peroxides — not HCl, not HI. The reason: only bromine radical is stable enough for the free radical chain mechanism to propagate. HCl and HI do not show the peroxide effect. The NTA has tested this specific distinction at least once in each of the last three papers.
Alkynes and Benzene — Electrophilic Substitution and Orientation Rules
From Alkynes, the NTA tests two things consistently:
- Acidic character of terminal alkynes — the sp-hybridised carbon holds the C−H bond electrons closer to the nucleus, making the hydrogen more acidic. Terminal alkynes react with sodium (Na) and sodamide (NaNH₂) to form sodium acetylide. The NTA tests this with: “Which compound reacts with NaNH₂?” — the answer is always the terminal alkyne.
- Hydration of alkynes — adding water across a triple bond gives an enol, which immediately tautomerises to a ketone (Markovnikov) or aldehyde (anti-Markovnikov using HgSO₄/H₂SO₄ for internal alkynes gives ketones; terminal alkynes give acetaldehyde as the exception).
From Benzene, the NTA tests two things consistently:
- Electrophilic Aromatic Substitution (EAS) — nitration, sulphonation, halogenation and Friedel-Crafts reactions. These are the four EAS reactions the NTA uses. Know the reagent for each.
- Ortho/Para vs Meta directing groups — this determines where the second substituent enters the ring.
| Directing Group | Type | Effect on Ring | Example |
| −OH, −NH₂, −OR, −CH₃ | Ortho/Para directors | Activate the ring (more reactive) | Phenol, Aniline |
| −NO₂, −CHO, −COOH, −CN | Meta directors | Deactivate the ring (less reactive) | Nitrobenzene |
| Halogens (−X) | Ortho/Para directors | Deactivate the ring | Chlorobenzene |
The most common trap: halogens are ortho/para directors (by resonance) but ring deactivators (by inductive effect). Students who only memorise “ortho/para = activating” lose marks when the NTA asks about reactivity. Halogens are the exception — they direct ortho/para but slow the ring down.
Haloalkanes and Haloarenes — SN1, SN2 and the Reagent Pairs That Confuse Students
Haloalkanes is one of those chapters where students feel confident after studying — and then lose marks they did not expect to lose. The reason is almost always the same: they understood SN1 and SN2 individually, but they never built a clear mental model for when each one kicks in.
The NTA does not ask you to define SN1. It gives you a substrate, a nucleophile and a solvent — and asks you to predict the product or identify the mechanism. That requires more than definitions. It requires decision-making.
SN1 vs SN2 — The Actual Difference and How NTA Tests It
Think of SN1 and SN2 as two completely different personalities that react to the same situation in opposite ways.
SN1 (Unimolecular Substitution) is a two-step reaction. The C−X bond breaks first to form a carbocation — then the nucleophile attacks. Because a carbocation intermediate forms, the reaction rate depends only on the substrate concentration (first order). The more stable the carbocation, the faster the SN1 reaction.
SN2 (Bimolecular Substitution) is a one-step reaction. The nucleophile attacks the carbon at the same moment the leaving group leaves — from exactly the opposite side. No carbocation forms. The rate depends on both the substrate and the nucleophile (second order). Steric hindrance is the enemy of SN2 — bulky groups around the carbon physically block the nucleophile from attacking.
| Feature | SN1 | SN2 |
| Steps | 2 (slow then fast) | 1 (concerted) |
| Intermediate | Carbocation | None (transition state only) |
| Best substrate | Tertiary > Secondary | Primary > Secondary |
| Solvent preferred | Polar protic (water, alcohol) | Polar aprotic (acetone, DMSO) |
| Stereochemistry | Racemisation (both sides attacked) | Inversion (Walden inversion) |
| Rate depends on | Substrate only | Substrate + Nucleophile |
| NTA tests | Substrate choice + stability | Stereochemistry + steric effect |
The trap the NTA uses most often: A secondary substrate with a strong nucleophile. Secondary substrates can go either way — SN1 or SN2 — and the deciding factor is the nucleophile strength and solvent. Strong nucleophile + polar aprotic solvent → SN2. Weak nucleophile + polar protic solvent → SN1.
One more thing the NTA tests specifically in SN1: carbocation rearrangement. When a secondary carbocation can rearrange to a more stable tertiary one via a hydride shift or methyl shift, it will — and the final product reflects the rearranged carbocation, not the original one. Students who do not account for rearrangement choose the wrong product every time.
Nucleophilic Substitution vs Elimination — When Does Each Happen?
When a haloalkane meets a base or nucleophile, two reactions compete: substitution (SN1/SN2) and elimination (E1/E2). The NTA tests exactly when each one wins.
The rule is clean and reliable:
- Strong base + high temperature → Elimination wins (E2 for primary/secondary, E1 for tertiary)
- Strong nucleophile + low temperature → Substitution wins
- Bulky base (like KOtBu — potassium tert-butoxide) → Always elimination, because the bulky base cannot reach the carbon to substitute — it pulls the β-hydrogen instead
- Aqueous KOH → Substitution (SN2 for primary)
- Alcoholic KOH → Elimination (E2)
Students who miss this distinction lose 1–2 questions per NEET paper on it. The alcoholic vs aqueous KOH distinction is one of the NTA’s favourite single-line traps — it has appeared in at least three of the last five papers.
Saytzeff’s Rule — when elimination gives two possible alkenes, the major product is the more substituted one (more alkyl groups on the double bond = more stable alkene).
Named reactions in this chapter to know for NEET:
- Finkelstein reaction — alkyl chloride + NaI (in acetone) → alkyl iodide
- Swarts reaction — alkyl chloride + AgF → alkyl fluoride
- Wurtz reaction — two alkyl halides + Na (dry ether) → higher alkane
Aldehydes, Ketones and Carboxylic Acids — The Highest-Yield Organic Chapter in NEET
This is the chapter that separates students who score 50+ in Chemistry from those who score 35. With 3–4 questions per paper — the highest single-chapter contribution in all of Organic Chemistry — it is also the chapter where the NTA is most creative with its traps.
The good news: almost every question from this chapter traces back to one mechanism — nucleophilic addition. Once you understand why nucleophiles attack the carbonyl carbon, you can predict what every reagent does, which product forms, and why aldehydes are more reactive than ketones.
Nucleophilic Addition Reactions — Which Reagents Give Which Products
The carbonyl carbon (C=O) is electron-deficient — the electronegative oxygen pulls electron density away, leaving the carbon partially positive and hungry for a nucleophile. This is why nucleophiles attack it. Every nucleophilic addition reaction in this chapter is a variation of this one theme.
Why aldehydes are more reactive than ketones in nucleophilic addition: Two reasons — one electronic, one steric. Alkyl groups are electron-donating (+I effect), which reduces the partial positive charge on the carbonyl carbon. Ketones have two alkyl groups doing this; aldehydes have only one. Additionally, two alkyl groups create more steric hindrance, making the nucleophile’s approach more difficult. Both effects make aldehydes more reactive.
The NTA tests this comparison directly — and it has appeared as a statement-based question in multiple papers.
| Reagent | Reacts With | Product | Key Point |
| NaBH₄ | Aldehydes + Ketones | Primary / Secondary alcohol | Mild reducing agent — does not reduce carboxylic acids |
| LiAlH₄ (LAH) | Aldehydes + Ketones + Carboxylic Acids | Primary / Secondary alcohol | Stronger — reduces everything including RCOOH |
| HCN | Aldehydes + Methyl ketones | Cyanohydrin | Addition across C=O |
| NH₂OH | Aldehydes + Ketones | Oxime (C=N−OH) | Nucleophile is N, not O |
| N₂H₄ | Aldehydes + Ketones | Hydrazone (C=N−NH₂) | Used in Wolff-Kishner |
| Grignard (RMgX) | Aldehydes + Ketones | 2° or 3° alcohol | HCHO gives 1° alcohol |
| Tollen’s reagent | Aldehydes only | Silver mirror | Distinguishes aldehyde from ketone |
| Fehling’s solution | Aliphatic aldehydes only | Red precipitate (Cu₂O) | Does NOT react with aromatic aldehydes or ketones |
The most commonly confused pair in NEET Chemistry: NaBH₄ vs LiAlH₄.
- NaBH₄ is selective — reduces only aldehydes and ketones, not carboxylic acids or esters
- LiAlH₄ is powerful — reduces everything (aldehydes, ketones, carboxylic acids, esters, amides)
- NTA trap: A question gives a molecule with both a ketone and a carboxylic acid group and asks which reagent gives which product — NaBH₄ reduces only the ketone; LiAlH₄ reduces both groups
Named Reactions in This Chapter — Aldol, Cannizzaro, Clemmensen, Wolff-Kishner
These are not just names to memorise — each one tests a specific condition or requirement. The NTA frames questions around the conditions, not just the names.
- Aldol Condensation
Aldehydes (or ketones) with α-hydrogen react with dilute NaOH to form a β-hydroxy aldehyde (aldol), which on heating eliminates water to give an α,β-unsaturated carbonyl compound.
- NTA condition tested: Must have an α-hydrogen. Benzaldehyde has NO α-hydrogen — it cannot give aldol condensation alone. It gives Crossed Aldol (Claisen-Schmidt) with acetaldehyde.
- NTA trap: “Which compound does NOT undergo aldol condensation?” → Answer: formaldehyde (HCHO) and benzaldehyde — both lack α-hydrogen.
- Cannizzaro Reaction
Aldehydes WITHOUT α-hydrogen undergo self-oxidation-reduction (disproportionation) in concentrated NaOH. One molecule of aldehyde gets oxidised to carboxylic acid; another gets reduced to alcohol.
- NTA key: Only works with aldehydes having NO α-hydrogen: HCHO, C₆H₅CHO, (CH₃)₃CCHO
- NTA trap: Students confuse Cannizzaro (no α-H, conc. NaOH) with Aldol (with α-H, dilute NaOH). The α-hydrogen is the deciding factor between the two.
- Clemmensen Reduction
Reduces the C=O of aldehydes and ketones directly to CH₂ using Zn-Hg amalgam in concentrated HCl. Used for acid-sensitive molecules.
- NTA tests: The reagent (Zn-Hg/conc. HCl) and the condition (acidic medium).
- Wolff-Kishner Reduction
Same result as Clemmensen (C=O → CH₂) but uses N₂H₄ (hydrazine) + KOH in ethylene glycol. Used for base-sensitive molecules.
- NTA trap: Both Clemmensen and Wolff-Kishner reduce C=O to CH₂ — the question tests WHICH reagent is used under WHICH conditions (acidic vs basic medium).
| Named Reaction | Reagent/Condition | Substrate Requirement | Product |
| Aldol Condensation | Dilute NaOH | Must have α-H | β-hydroxy carbonyl → α,β-unsaturated carbonyl |
| Cannizzaro Reaction | Conc. NaOH | No α-H | Alcohol + Carboxylate salt |
| Clemmensen Reduction | Zn-Hg / conc. HCl | Aldehyde or Ketone | C=O → CH₂ (acidic conditions) |
| Wolff-Kishner Reduction | N₂H₄ + KOH / ethylene glycol | Aldehyde or Ketone | C=O → CH₂ (basic conditions) |
| Rosenmund Reduction | H₂ / Pd-BaSO₄ | Acyl chloride (RCOCl) | Aldehyde (RCHO) |
| Hell-Volhard-Zelinsky | Br₂ + red P | Carboxylic acid | α-bromo acid |
Amines and Biomolecules — Small Chapters, Consistent Questions
These two chapters sit at the end of Class 12 Organic Chemistry — and students often reach them exhausted, revise them lightly and lose 2–3 marks they could have easily kept. Together they contribute 2–4 questions per paper. The NTA draws from a narrow, predictable set of concepts in both — which means a focused 2-day revision of each is all you need.
Amines — Basicity Order, Reactions and the Gabriel Synthesis
The most tested concept in Amines is not a reaction — it is the basicity order. The NTA has asked this in some form in almost every paper from 2019 to 2025.
Basicity order in aqueous solution:
Aliphatic secondary amine > aliphatic primary amine > ammonia > aromatic amine (aniline)
Written more specifically: (CH₃)₂NH > CH₃NH₂ > NH₃ > C₆H₅NH₂
Here is why each step happens:
- Aliphatic amines are more basic than ammonia because alkyl groups donate electrons (+I effect), increasing electron density on nitrogen and making it a better proton acceptor
- Secondary amines are slightly more basic than primary amines in aqueous solution — because the extra alkyl group donates more electron density, and water stabilises the secondary ammonium ion well through solvation
- Aniline is far weaker than aliphatic amines — the lone pair on nitrogen is delocalised into the benzene ring through resonance, making it far less available for protonation
The NTA trap on basicity: Students often rank tertiary aliphatic amines as most basic because they have three alkyl groups. In gas phase, this would be correct. In aqueous solution, tertiary ammonium ions are poorly solvated — bulky alkyl groups prevent water molecules from stabilising the ion, so the actual measured basicity in water follows the order above. The NTA tests aqueous basicity — always specify the medium.
Reactions the NTA tests in Amines:
| Reaction | Reagent/Condition | Substrate | Product | NTA Test Focus |
| Gabriel Synthesis | Phthalimide + KOH + RX → then acid hydrolysis | — | Primary aliphatic amine only | “Which method gives only primary amine?” |
| Hoffmann Bromamide | Br₂ + KOH | Amide (RCONH₂) | Primary amine with one less carbon | Carbon chain decreases by 1 |
| Carbylamine reaction | CHCl₃ + KOH | Primary amine only | Isocyanide (foul smell) | Test for primary amine — does NOT work for 2° or 3° |
| Diazotisation | NaNO₂ + HCl (0–5°C) | Primary aromatic amine | Diazonium salt | Low temperature is essential |
| Coupling reaction | Diazonium salt + phenol/aniline | Diazonium salt | Azo dye (orange/yellow) | Used to identify aromatic amines |
The carbylamine reaction is the NTA’s favourite Amines test. It appears in at least one paper every two years — asking either which amine gives the carbylamine test (only primary amines) or which reagent is used (CHCl₃ + alc. KOH).
Biomolecules — What NEET Tests From Carbohydrates, Proteins and Nucleic Acids
Biomolecules contributes 1–2 questions per paper — and the NTA has a very short list of things it actually tests. Students who revise the entire chapter lose time. Students who focus on the following points cover everything the NTA draws from:
From Carbohydrates:
- Glucose is an aldohexose. Fructose is a ketohexose. Both are C₆H₁₂O₆ but structural isomers.
- Reducing sugars: all monosaccharides + maltose + lactose. Non-reducing sugar: sucrose (both anomeric carbons involved in glycosidic bond — no free −CHO or −OH to reduce Tollens/Fehling)
- Starch = amylose (unbranched, α-1,4 glycosidic bonds) + amylopectin (branched, α-1,4 and α-1,6 bonds)
- Cellulose = β-1,4 glycosidic bonds (humans cannot digest because we lack β-glucosidase)
From Proteins:
- Four structural levels: Primary (peptide bond sequence) → Secondary (α-helix/β-sheet via hydrogen bonds) → Tertiary (3D shape via disulphide, hydrophobic, ionic bonds) → Quaternary (multiple polypeptide chains)
- Denaturation = loss of 3D structure (secondary/tertiary) — primary structure (peptide bonds) is NOT broken
- The NTA regularly asks: “Which bonds are broken during denaturation?” → Answer: hydrogen bonds, disulphide bonds — NOT peptide bonds
From Nucleic Acids:
- DNA is double-stranded; RNA is single-stranded
- DNA has deoxyribose sugar and thymine; RNA has ribose sugar and uracil
- Chargaff’s rule applies to DNA only — A=T, G=C
- The NTA tests the base-pairing rules and the sugar difference in single-statement questions
How to Actually Study Organic Chemistry for NEET — Mechanism Over Memorisation
Here is a fact from recent NEET data: students who memorise reactions score 20–35 marks in Organic Chemistry. Students who understand mechanisms score 40–50 marks. The difference — roughly 15 marks — can be the difference between getting MBBS and not. That gap is not talent. It is method.
The method that works is simple to describe and takes discipline to follow: learn the mechanism first, then the reaction, then the product. Not the other way around.
The 4 Core Mechanisms That Predict 90% of NEET Organic Reactions
Every Organic Chemistry question in NEET — across all chapters — traces back to one of these four mechanisms. Once you know them, you stop memorising individual reactions and start predicting them.
- Nucleophilic Substitution (SN1/SN2)
A nucleophile (electron-rich) attacks an electron-deficient carbon — replacing the leaving group. Covers: all of Haloalkanes, some of Alcohols, some of Carbonyl reactions.
Ask yourself: Is there a good leaving group? Is the nucleophile strong or weak? What is the substrate — primary, secondary or tertiary? - Electrophilic Addition
An electrophile attacks a pi bond (alkene, alkyne). Covers: all Hydrocarbon addition reactions, Markovnikov/Anti-Markovnikov, ozonolysis.
Ask yourself: Which carbon is more electron-rich? That is where the electrophile goes last — and the nucleophile goes first. - Nucleophilic Addition to C=O
A nucleophile attacks the partially positive carbonyl carbon. Covers: all of Aldehydes and Ketones, Grignard reactions, reduction reactions.
Ask yourself: Is this a C=O compound? If yes, the nucleophile attacks carbon. Which nucleophile? That tells you the product. - Electrophilic Aromatic Substitution (EAS)
An electrophile attacks the electron-rich benzene ring. Covers: all Benzene reactions (nitration, sulphonation, Friedel-Crafts), directing effects.
Ask yourself: What is already on the ring? That tells you where the next group goes — and whether the ring is activated or deactivated.
How to Build a Reaction Map — The Revision System That Replaces Rote Learning
A reaction map is a one-page visual that connects functional groups through reagents and conditions. It answers the most common NTA question type: “What is the product when compound X reacts with reagent Y?” — without requiring you to recall a list.
How to build one (takes 45 minutes per chapter):
- Write the parent functional group in the centre (e.g., Alkene for Hydrocarbons)
- Draw arrows outward — one arrow per reaction
- Label each arrow with the reagent + condition (not just the reagent name)
- Write the product at the tip of each arrow
- Colour-code by mechanism type — all nucleophilic additions in blue, all eliminations in red, all EAS in green
Once built, your revision takes 5 minutes per chapter — you trace the arrows instead of re-reading pages. Students who build reaction maps in Weeks 1–2 revise 3× faster in Weeks 3–4 than students who use notes.
Revision sequence for Organic Chemistry (follow in this order):
GOC → Hydrocarbons → Haloalkanes → Alcohols, Phenols and Ethers → Aldehydes, Ketones and Carboxylic Acids → Amines → Biomolecules → Polymers
This is not alphabetical and not the textbook order — it is the dependency order. Each chapter uses concepts from the one before it. Reversing the sequence creates confusion; following it creates momentum.
The 7 Organic Chemistry Mistakes That Cost Students 20+ Marks
These are not general study mistakes. These are specific Organic Chemistry errors that show up in mock analysis again and again — and they are entirely avoidable once you know what to look for.
- Treating reactions as a list instead of a pattern.
Students who memorise 60 reactions individually forget them under pressure. Students who connect reactions to the 4 mechanisms remember them because they understand them. If you are still using a “reactions list” in Week 3, switch to the reaction map approach in Section 7 now — not later. - Confusing NaBH₄ with LiAlH₄.
NaBH₄ reduces only aldehydes and ketones. LiAlH₄ reduces everything including carboxylic acids and esters. The NTA has used this exact confusion as a trap in consecutive papers. When you see a molecule with multiple functional groups and a reducing agent — stop and identify exactly which groups each reagent can and cannot reduce. - Applying SN1 to primary substrates or SN2 to tertiary substrates.
SN2 needs an unhindered primary carbon. SN1 needs a stable carbocation (tertiary preferred). Students who mix these up choose the wrong mechanism and the wrong product. Before answering any substitution question — identify the substrate class first. - Missing the α-hydrogen rule in Aldehyde/Ketone reactions.
Aldol condensation requires α-hydrogen. Cannizzaro reaction requires no α-hydrogen. If you do not check for α-hydrogen before assigning the reaction type, you will choose the wrong product every time. Make this a reflex — see a carbonyl compound, immediately check for α-H. - Forgetting that the carbylamine test is only for primary amines.
The NTA tests this annually. Secondary and tertiary amines do not give the carbylamine test. When a question asks which amine reacts with CHCl₃/KOH — the answer is always the primary amine only. - Skipping stereochemistry in Haloalkane questions.
SN2 gives inversion of configuration (Walden inversion). SN1 gives racemisation. Students who know the mechanism but forget the stereochemical outcome lose marks on questions that ask for the product’s optical activity. Spend 30 minutes specifically on stereochemistry in Week 2 — it covers 1–2 questions per paper. - Using Morrison & Boyd or other reference books when NCERT has the answer.
NEET Organic Chemistry is based entirely on NCERT Class 11 and 12. Every reaction, every named reaction and every exception in this guide traces to a specific NCERT page. Students who go to reference books for Organic Chemistry waste 3–4 weeks covering content the NTA will never test.
Frequently Asked Questions — NEET Organic Chemistry 2026
Is NCERT enough for NEET Organic Chemistry 2026?
Yes — NCERT is enough for NEET Organic Chemistry, provided you read it with mechanism understanding and not just as a reactions list. The NTA draws every named reaction, every reagent and every exception directly from NCERT Class 11 and 12 Chemistry. Where students go wrong is reading NCERT passively — they see the reaction table, think they know it and move on without testing whether they can predict the product independently. One pass of NCERT with active recall — covering the page and writing the product from memory — covers everything the NTA tests in Organic Chemistry.
How many questions come from Organic Chemistry in NEET?
Organic Chemistry contributes approximately 13–15 questions out of 45 Chemistry questions — roughly 34% of the Chemistry section, or 52–60 marks out of 180. Aldehydes, Ketones and Carboxylic Acids is the single highest-yield chapter (3–4 questions), followed by GOC and Hydrocarbons (2–3 each). Together these three chapters generate 7–10 questions — more than half of the entire Organic Chemistry section.
What are the most important named reactions for NEET Organic Chemistry?
The 8 named reactions that appear most consistently across NEET papers are: Aldol Condensation, Cannizzaro Reaction, Clemmensen Reduction, Wolff-Kishner Reduction, Rosenmund Reduction, Gabriel Synthesis, Hoffmann Bromamide Degradation and Carbylamine Reaction. For each one, know three things: the substrate it requires, the reagent/condition, and the product. If you know these three for all 8, you will answer every named reaction question correctly.
How do I remember organic reactions without rote memorisation?
The most effective method is building a reaction map for each chapter — a one-page visual connecting functional groups through reagents and products. Pair this with mechanism understanding: once you know that nucleophiles attack electron-deficient carbons, you stop memorising individual reactions and start predicting them. Students who switch from memorisation to mechanism-based learning typically improve their Organic Chemistry score by 10–15 marks within 3–4 weeks of consistent practice.
How long does it take to complete Organic Chemistry for NEET?
A realistic timeline for thorough first-time coverage of all 8 Organic Chemistry chapters is 25–35 days at 3–4 hours per day — with GOC taking the longest (4–5 days) because everything else depends on it. Revision after the first pass takes 3–4 days per chapter cycle. Students who start Organic Chemistry with GOC and follow the dependency order (GOC → Hydrocarbons → Haloalkanes → Alcohols → Carbonyls → Amines → Biomolecules → Polymers) consistently finish faster and retain better than those who follow textbook chapter order.
Conclusion
Organic Chemistry in NEET is 52–60 marks. You have just covered the mechanism logic, the chapter-wise reactions, the named reaction conditions and the 7 mistakes that silently drain scores. The framework is here — what turns it into marks is consistent practice against actual NTA question patterns.
If you find that identifying which mechanism applies, analysing mock errors in Chemistry or staying on track across all three subjects is where your preparation slips — that is exactly what the
at EduAiTutors addresses. Week 2 is a dedicated Chemistry Power Week — live sessions on Organic mechanisms, reagent pair drilling and AI-powered mock analysis that flags your specific Organic Chemistry gaps after every test.
The reactions are learnable. The marks are available. Use both.
