Inorganic Chemistry for NEET and JEE 2026: The Complete Expert Guide to Scoring Full Marks

Here is something I tell every student who walks into my class looking stressed about Chemistry: the students who score 160 out of 180 in NEET Chemistry are not the ones who studied the most hours. They are the ones who studied Inorganic Chemistry the right way while everyone else was avoiding it.
Inorganic Chemistry for NEET and JEE 2026 contributes somewhere between 10 and 13 questions in NEET Chemistry alone. That is 40 to 52 marks sitting right there, fully available to any student willing to understand the logic behind the content rather than fighting a losing battle with blind memorisation.
In 15 years of teaching, I have mentored students who scored zero in Inorganic and students who scored full marks from it. The difference between those two groups was never intelligence. It was always strategy. This guide gives you that strategy, chapter by chapter, concept by concept, exactly the way I teach it in my classroom.
NEET 2026 Last 30 Days Timetable Realistic 4-Week Plan That Actually Works
Why Most Students Struggle With Inorganic Chemistry and Why You Will Not
The honest answer to this question is simple. Most students struggle because they approach Inorganic Chemistry as a collection of disconnected facts to memorise. When you approach it that way, you are fighting an unwinnable battle. There are thousands of individual pieces of information spread across eight chapters. No human brain retains thousands of disconnected facts under exam pressure.
The students who score well do not have better memories. They have a better system.
The Three Root Causes of Poor Inorganic Scores
Root Cause 1: Treating it like rote learning from Day One
When a student opens the p-Block chapter, sees pages of reactions and properties and exceptions, and immediately decides “I will memorise all of this,” they have already lost. Memorisation without understanding creates fragile, short-term recall. The exam is designed to test whether you understand why something happens, not just that it happens.
Root Cause 2: Leaving it for the last month
Inorganic Chemistry is the chapter where the 30th revision is worth more than the 1st revision. Every time you revisit NCERT, you pick up details you missed before. Students who start Inorganic in the final month get one rushed read and walk into the exam with 40% retention. Students who start early and revise repeatedly walk in with 90% retention.
Root Cause 3: Ignoring exceptions
The NTA loves exceptions. The element that does not follow its group trend. The compound that behaves differently from its family. The reaction that surprises you. These exceptions appear in NEET and JEE Main papers year after year, and students who have not specifically noted and understood them lose these marks every single time.
The Mental Shift That Changes Everything
Here is the framework I give every student before we begin Inorganic Chemistry together.
Every element’s behaviour follows from its electronic configuration. Every periodic trend follows from how atomic size, nuclear charge and electron shielding change across and down the periodic table. Every reaction follows from the stability preferences of the atoms involved. Every exception exists because of a specific, understandable reason.
When you study with this lens, you are not memorising facts. You are understanding a system. And understanding a system is five times faster to build and ten times more durable under exam pressure than a list of memorised facts. We are going to build that understanding together, chapter by chapter, right now. Read More: NEET Dropper Mental Health — The Complete Guide to Managing Anxiety, Burnout and Depression During Your Drop Year
Inorganic Chemistry Chapter-wise Weightage for NEET and JEE 2026
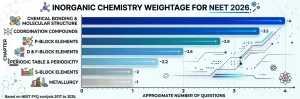
Before building any strategy, you need to know where the marks actually live. Here is the picture based on careful analysis of NEET and JEE Main papers from 2017 to 2025.
Where the Maximum Marks Are
The Chapters Students Skip and Regret Every Year
Every year, two chapters get systematically skipped by students running short on time: s-Block Elements and Hydrogen. Students assume these are minor chapters not worth the investment.
Here is the reality. s-Block and Hydrogen together give you 2 to 3 reliable questions in NEET. You can complete both chapters in 4 to 5 focused days of NCERT reading. That is the best marks-per-day ratio in all of Inorganic Chemistry. Students who skip them leave guaranteed marks on the table. Do not be that student.
NEET 2026 Genetics and Evolution All Concepts Diagrams and 50 Most Tested Questions
Chemical Bonding and Molecular Structure: The Highest Weightage Chapter in Inorganic Chemistry for NEET and JEE
Chemical Bonding is where Inorganic Chemistry should always begin. With 3 to 4 questions in NEET and 2 to 3 in JEE Main appearing almost every year, this chapter alone can determine whether your Chemistry score is good or exceptional.
The good news is that Chemical Bonding is the most logical chapter in all of Inorganic Chemistry. Nothing here is arbitrary. Every concept follows from the structure of atoms and how they interact. Let me walk you through every testable concept the way I would explain it sitting with you in my classroom.
Hybridisation Made Simple With the One Rule That Covers Everything
Hybridisation confuses students because they try to memorise which molecule has which hybridisation. That approach breaks down the moment the exam shows them a molecule they have not seen before.
Here is the one rule that covers every molecule you will encounter in NEET and JEE:
Hybridisation = Number of bond pairs on the central atom + Number of lone pairs on the central atom
-
Answer is 2: sp hybridisation, linear electron geometry, bond angle 180 degrees
-
Answer is 3: sp2 hybridisation, trigonal planar electron geometry, bond angle 120 degrees
-
Answer is 4: sp3 hybridisation, tetrahedral electron geometry, bond angle 109.5 degrees
-
Answer is 5: sp3d hybridisation, trigonal bipyramidal electron geometry
-
Answer is 6: sp3d2 hybridisation, octahedral electron geometry
You can derive the hybridisation of any molecule in under 10 seconds using this rule. You never need to memorise individual molecules again.
Now here is the part where most students lose marks. They calculate hybridisation correctly but write the wrong molecular shape. This is because hybridisation gives you the electron pair geometry, not the molecular shape. The molecular shape only considers the positions of atoms, not lone pairs.
Water is the classic example. sp3 hybridisation means 4 electron pairs arranged tetrahedrally. But two of those four positions are occupied by lone pairs. Remove the lone pairs from the shape description and you get a bent or V-shaped molecule, not a tetrahedral one. Bond angle drops from 109.5 degrees to 104.5 degrees because lone pair-lone pair repulsion is stronger than bond pair-bond pair repulsion and pushes the bond pairs closer together.
This single distinction between electron geometry and molecular shape is responsible for more lost marks in Chemical Bonding than almost any other concept. Now you know it, and you will not make that mistake.
The complete VSEPR table you must know:
Understand every entry in this table rather than simply memorising it. If you understand why XeF4 is square planar and not octahedral, you can answer any shape question the exam throws at you.
VSEPR Theory and Why Bond Angles Are What They Are
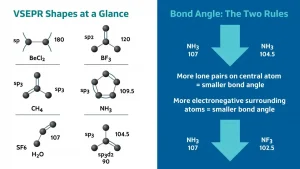
VSEPR stands for Valence Shell Electron Pair Repulsion. The core idea is beautifully simple: electron pairs around an atom repel each other and arrange themselves as far apart as possible to minimise repulsion. The shape that achieves this maximum separation is the shape the molecule adopts.
The repulsion order that drives all bond angle comparisons is:
Lone pair vs Lone pair repulsion is greater than Lone pair vs Bond pair repulsion, which is greater than Bond pair vs Bond pair repulsion
This single hierarchy explains every bond angle deviation from the ideal value:
-
NH3 has 107 degrees instead of 109.5 degrees because one lone pair pushes the three bond pairs slightly closer
-
H2O has 104.5 degrees instead of 109.5 degrees because two lone pairs push the two bond pairs even closer
-
NF3 has a smaller bond angle than NH3 despite having the same hybridisation because fluorine is highly electronegative and pulls bonding electron pairs toward itself and away from nitrogen, reducing repulsion between bond pairs and allowing them to come closer
That last point about electronegativity is where marks hide in both NEET and JEE. Students who know only the lone pair rule get the NH3 vs NF3 comparison wrong. You now know both rules.
Quick Tip From Faculty:
When a question asks you to compare bond angles of two similar molecules, work through these two checks in order:
First check: Which molecule has more lone pairs on the central atom? More lone pairs means smaller bond angle.
Second check: Which molecule has more electronegative atoms surrounding the central atom? More electronegative surrounding atoms pull bond pairs away from the centre, reducing bond pair repulsion, which allows the bond angle to decrease.
Apply both checks and you will answer every bond angle comparison question correctly. I have seen this approach work for hundreds of students who previously struggled with these questions. This is where most students lose marks in Chemical Bonding, but not you, because now you have the complete framework.
Molecular Orbital Theory: What Each Exam Actually Tests
For NEET, you need a working understanding of bond order calculation and the magnetic properties of simple diatomic molecules. For JEE Main, you need that plus the ability to write complete MO electron configurations. Let me give you exactly what each exam needs.
The Bond Order Formula:
Bond Order = (Electrons in bonding MOs minus Electrons in antibonding MOs) divided by 2
Bond order of zero means the molecule does not exist in a stable form. Bond order of 1, 2 or 3 means single, double or triple bond respectively.
The MO filling sequence for N2 and lighter molecules:
σ1s, σ1s, σ2s, σ2s, π2px = π2py, σ2pz, π2px = π2py, σ*2pz
The MO filling sequence for O2 and heavier molecules:
σ1s, σ1s, σ2s, σ2s, σ2pz, π2px = π2py, π2px = π2py, σ*2pz
Notice that the position of σ2pz and π2p swaps between N2 and O2. This switch matters enormously because it affects the magnetic properties of these molecules.
The facts you must know for NEET and JEE:
-
O2 is paramagnetic. This surprises many students because the Lewis structure of O2 shows all electrons paired. MOT reveals that O2 has 2 unpaired electrons in the π*2p orbitals and is therefore paramagnetic. This is a direct NEET PYQ.
-
N2 has a bond order of 3 and is diamagnetic.
-
B2 is paramagnetic with bond order 1 and 2 unpaired electrons in π2p orbitals.
-
He2 has bond order 0 and does not exist as a stable molecule.
-
NO has bond order 2.5 and is paramagnetic.
For JEE specifically: Know how to derive bond orders and magnetic properties of molecular ions too. O2+, O2-, N2+ and N2- all appear in JEE Main papers. Remove or add electrons from the highest occupied MO in the standard filling sequence and recalculate bond order.
Common Mistakes Students Make in Chemical Bonding
Mistake 1: Confusing electron geometry with molecular shape
Writing that H2O is tetrahedral because it has sp3 hybridisation loses you the mark every time. The molecular shape is bent. Hybridisation describes the electron arrangement. Molecular shape describes only the atom positions.
Mistake 2: Applying only the lone pair rule for bond angle comparisons
NH3 and NF3 have the same hybridisation and the same number of lone pairs. Their bond angles are different because of the electronegativity effect. Students who know only the lone pair rule get this wrong.
Mistake 3: Using Lewis structures to determine magnetic properties
Lewis structures cannot show you unpaired electrons accurately in all cases. The O2 paramagnetism question is the most famous example. Always use MOT for paramagnetism or diamagnetism questions.
Mistake 4: Getting the MO filling order wrong for O2 versus N2
The swap in the position of σ2pz and π2p happens between N2 and O2. Students who memorise only one sequence get the electronic configuration of O2 and F2 wrong. Remember: the swap happens at O2.
Mistake 5: Forgetting about expanded octets in hybridisation calculations
Phosphorus and sulphur can accommodate more than 8 electrons because they have available d orbitals. PCl5, SF6 and IF7 all have expanded octets. Students who apply the octet rule to these molecules get the hybridisation and shape wrong.
Types Of Previous Year Question Analysis: Chemical Bonding
These are the five concept types that appear most consistently in NEET and JEE Main from this chapter. If you can answer all five types confidently, you are scoring full marks from Chemical Bonding.
1: Shape and hybridisation of molecules with lone pairs or expanded octets
Every year, the molecule chosen for this question type is one that either has lone pairs on the central atom or has an expanded octet. The exam never tests the shape of CH4 because every student knows it. It tests XeF4, ClF3, PCl5, SF6 and similar molecules. Master every molecule in the VSEPR table above and this question type becomes automatic.
2: Bond order and bond length comparisons using MOT
“Arrange N2, N2+ and N2- in increasing order of bond length.” These questions require you to calculate bond order from MOT, then use the relationship that higher bond order equals shorter and stronger bond. Bond length and bond order always move in opposite directions.
3: Paramagnetism or diamagnetism of a diatomic species
Any question about magnetic properties of O2, B2, NO or their ions is an MOT question. Write the complete electron configuration, count unpaired electrons and give your answer. O2 being paramagnetic appears in NEET papers in different forms regularly.
4: Bond angle comparison between similar molecules
“Which has a larger bond angle, NH3 or NF3?” Apply the two-check system from the Faculty Tip above. These questions appear more frequently in JEE than in NEET but do appear in both.
5: Formal charge calculation
JEE papers test formal charges on atoms in resonance structures. The formula is: Formal Charge equals Valence electrons minus Non-bonding electrons minus half of Bonding electrons. The resonance structure with the lowest formal charges and negative formal charges on the most electronegative atom is the most stable contributing structure.
Quick Tip From Faculty: Chemical Bonding PYQ Pattern
After analysing every NEET paper from 2015 to 2025, here is what I can tell you with complete confidence. The Chemical Bonding questions in NEET almost always target one of these three things: molecules where the molecular shape is different from the electron geometry because of lone pairs, molecules where an exception exists to what the student expects, and MOT-based magnetic property questions.
Study every exception in Chemical Bonding with its reason and you will be prepared for exactly what the exam sets. This is not an accident. Paper setters specifically choose questions that test whether students understand the why, not just the what. Understanding the why is exactly what this section has given you.
NEET Biology Chapter-wise Previous Year Questions With Answers and Explanations
Periodic Table and Periodic Trends: Where Free Marks Are Hiding in Inorganic Chemistry for NEET and JEE
If I had to pick one chapter in Inorganic Chemistry where prepared students consistently outscore unprepared students by the widest margin, it would be Periodic Table and Periodic Trends. The questions from this chapter are direct, predictable and repeat year after year.
The entire chapter comes down to six trends and about ten exceptions. Master those sixteen things and you will answer every periodic trends question correctly every single time.
The Six Periodic Trends Every Student Must Know Cold
Trend 1: Atomic Radius
Atomic radius decreases across a period from left to right and increases down a group from top to bottom.
Across a period: nuclear charge increases as protons are added, but electrons are added to the same shell. The stronger nuclear pull drags the electron cloud inward. Each element from left to right in a period is slightly smaller than the one before it.
Down a group: new electron shells are added with each period. Each new shell is farther from the nucleus. Inner shells also shield the outer electrons from the full nuclear attraction through the shielding effect. The result is a larger, more loosely held outer shell.
The exception you must know: Gallium (Ga) has almost the same atomic radius as Aluminium (Al) despite being one full period below it. This happens because of the d-block contraction. The 10 d-electrons added between Al and Ga in Period 4 do not shield the outer electrons effectively. Nuclear charge increases more than expected, pulling the electron cloud inward and making Ga unexpectedly small. The same logic applies to the lanthanide contraction in the f-block.
Trend 2: Ionisation Enthalpy
Ionisation enthalpy is the energy required to remove the outermost electron from a gaseous atom. It increases across a period and decreases down a group.
Across a period: smaller atomic radius means the outermost electron sits closer to the nucleus and is more strongly attracted. More energy is needed to pull it away.
Down a group: larger atomic radius, stronger shielding, and a more loosely held outer electron means less energy is needed to remove it.
The two exceptions that appear in NEET and JEE papers every single year without fail:
Exception 1: The ionisation enthalpy of Beryllium (Be) is greater than that of Boron (B), even though B comes after Be in Period 2. The general trend says IE should increase from Be to B. But Be has a completely filled 2s orbital (2s2), which is extra stable. B has one electron in the 2p orbital (2s2 2p1), which is higher in energy and less stable. It is actually easier to remove the 2p electron from B than the 2s electron from Be.
Exception 2: The ionisation enthalpy of Nitrogen (N) is greater than that of Oxygen (O), even though O comes after N. N has a half-filled 2p subshell (2p3, one electron in each orbital), which is extra stable due to exchange energy. O has 2p4, meaning one 2p orbital holds a paired electron. That paired electron experiences extra repulsion from its partner and is easier to remove. So O has a lower IE than N despite coming later in the period.
These two exceptions, Be greater than B and N greater than O, appear in NEET papers and JEE Main papers with remarkable regularity. Know them cold. Know the reasons behind them.
Quick Tip From Faculty: Ionisation Enthalpy Exceptions
Here is the mental shortcut I give every student for remembering ionisation enthalpy exceptions. Think of it as the “extra stability resistance rule.” Completely filled subshells and half-filled subshells are extra stable configurations. Atoms with these configurations resist losing electrons more than the general trend predicts. Any time you see a question comparing the IE of two adjacent elements in Period 2, check first whether either element has a completely filled or half-filled subshell. If yes, that element has a higher IE than the general trend would suggest.
Trend 3: Electron Gain Enthalpy
Electron gain enthalpy is the energy change when a neutral gaseous atom accepts one electron. A more negative value means the atom more readily accepts an electron.
Generally, electron gain enthalpy becomes more negative across a period and less negative down a group.
The exception every student must know: Chlorine has a more negative electron gain enthalpy than Fluorine, despite F being above Cl in Group 17. You would logically expect F to accept electrons more readily because it is smaller and its nucleus is closer to the incoming electron. But F is so small that its 2p orbital is already compact and crowded. Adding an electron causes significant electron-electron repulsion in that compact space. Cl is larger, with 3p orbitals that accommodate the incoming electron more comfortably. So Cl wins here.
This is one of the most tested exceptions in all of Inorganic Chemistry. It appears not just in direct questions but also as the basis for explaining why F2 is a stronger oxidising agent than its electron gain enthalpy alone would suggest.
Trend 4: Electronegativity
Electronegativity measures an atom’s ability to attract the shared electrons in a bond toward itself. It increases across a period and decreases down a group.
Fluorine is the most electronegative element in the entire periodic table. This is a non-negotiable fact with direct consequences in bond polarity, hydrogen bonding, oxoacid strength comparisons and halogen reactivity questions throughout Inorganic Chemistry.
Trend 5: Metallic and Non-Metallic Character
Metallic character decreases across a period and increases down a group. Non-metallic character does exactly the opposite.
The diagonal line separating metals from non-metals passes through B, Si, Ge, As, Sb and Te. These elements, called metalloids or semimetals, show properties of both metals and non-metals. Germanium and Silicon being semiconductors is a direct application of this.
Trend 6: Oxidising and Reducing Power
Among non-metals, oxidising power increases across a period and decreases down a group. Fluorine is the strongest oxidising agent of all elements.
Among metals, reducing power increases down a group and decreases across a period. Caesium and Francium are the strongest reducing agents among the alkali metals.
The Exceptions the Exam Loves to Test
Collect all critical exceptions in one place for your revision list:
- F has lower electron gain enthalpy than Cl because of its compact 2p orbital and the resulting repulsion with the incoming electron
- IE of Be is greater than B because of Be’s extra stable fully filled 2s orbital
- IE of N is greater than O because of N’s extra stable half-filled 2p subshell
- Ga has unexpectedly small atomic radius due to poor shielding by d-electrons in Period 4
- Noble gases have positive electron gain enthalpy because their completely filled shells strongly resist accepting additional electrons
- The second ionisation enthalpy of Na is enormously high because removing the first electron gives Na+ the stable noble gas configuration of Neon, and the second electron must come from an inner shell
Common Mistakes in Periodic Trends
Mistake 1: Confusing electron gain enthalpy with electronegativity
Both relate to an atom’s attraction for electrons but they are different quantities. Electronegativity applies to atoms in bonds. Electron gain enthalpy applies to isolated gaseous atoms. The F versus Cl exception applies to electron gain enthalpy, not electronegativity.
Mistake 2: Ignoring noble gases in electron gain enthalpy questions
When a question asks about the trend in electron gain enthalpy across Period 2, it may go all the way to Neon. Ne has a positive electron gain enthalpy that breaks the general trend. Students who answer without checking where the period ends get this wrong.
Mistake 3: Applying the IE trend without checking for special configurations
The Be-B and N-O exceptions trip up students who apply the general rule mechanically. Always check for fully filled and half-filled subshells before comparing IE of adjacent elements.
NEET 2026 Stress Management A Practical Mental Health Guide for Every Stage of Preparation
s-Block Elements: Small Chapter, Reliable Marks in Every Paper
The s-Block elements are Group 1 (Li, Na, K, Rb, Cs, Fr) and Group 2 (Be, Mg, Ca, Sr, Ba, Ra). This is the chapter most students underestimate. It is short enough to finish completely in 4 to 5 days of focused NCERT reading, and it delivers 2 to 3 reliable questions in NEET every year.
The key to scoring full marks from s-Block is understanding the anomalous behaviour of Li and Be and the diagonal relationships they form. These are the concepts the exam tests, not the general group trends that every student already knows.
Key Reactions and Anomalous Behaviour
Reaction of Alkali Metals With Water
All Group 1 metals react with water to form a metal hydroxide and hydrogen gas. Reactivity increases sharply down the group: Li reacts gently, Na reacts vigorously with sparking, K reacts so violently the hydrogen ignites with a lilac flame, Rb and Cs react explosively.
2Na + 2H2O gives 2NaOH + H2
Reaction of Alkali Metals With Oxygen: The Most Tested s-Block Concept
This is where the group surprises students. Each alkali metal forms a different type of oxide when reacting with excess oxygen:
-
Li forms a normal oxide: 4Li + O2 gives 2Li2O
-
Na forms sodium peroxide: 2Na + O2 gives Na2O2
-
K, Rb and Cs form superoxides: K + O2 gives KO2
Why does this happen? The size-stability rule. Larger metal cations stabilise larger anions more effectively because of lattice energy considerations. The oxide ion O2- is small. The peroxide ion O22- is larger. The superoxide ion O2- is the largest. Li, the smallest alkali metal cation, can only stabilise the small oxide ion. As you go down the group and the cation gets larger, it can stabilise increasingly large anions.
This size-stability rule comes up across multiple Inorganic Chemistry chapters. Understanding it here gives you a tool you can use in coordination chemistry, halide stability comparisons and more.
Anomalous Behaviour of Lithium
Lithium behaves differently from the rest of Group 1 in several specific ways. All of these differences trace back to one root cause: Li+ has an unusually small ionic radius for a Group 1 cation, giving it an exceptionally high charge density.
The anomalous properties of Li that are directly tested in NEET:
- Li forms a nitride (Li3N) directly by reacting with N2 at room temperature. No other Group 1 metal does this.
- Li forms a normal oxide with O2, not a peroxide or superoxide.
- Li has the highest melting point in Group 1 because its small size allows closer packing.
- LiCl is soluble in ethanol and other organic solvents because the Li-Cl bond has significant covalent character due to Li’s high charge density.
- Li has an anomalously high hydration enthalpy despite being the lightest alkali metal, because its tiny ion attracts water molecules very strongly.
The Diagonal Relationship: Li Resembles Mg More Than Na
Because Li+ and Mg2+ have very similar charge densities despite being in different groups, their chemistry shows remarkable similarities. This diagonal relationship between Li and Mg is tested directly in NEET.
Similarities between Li and Mg:
- Both form normal oxides (not peroxides or superoxides)
- Both form nitrides directly with N2
- Both form carbonates that decompose on heating (Na2CO3 does not decompose easily)
- Both LiCl and MgCl2 are soluble in ethanol
- Both have high hydration enthalpies
- Both Li and Mg show high electronegativity compared to other members of their groups
Anomalous Behaviour of Beryllium
Be shows a similar pattern. Its small size and high charge density make its chemistry resemble Al in Period 3 more than it resembles Mg in its own group.
The anomalous properties of Be tested in NEET and JEE:
- Be does not react with water even at high temperatures (unlike Mg, Ca, Sr, Ba which all do)
- BeO and Be(OH)2 are amphoteric: they dissolve in both acids and bases. All other Group 2 oxides and hydroxides are purely basic.
- Be forms covalent compounds because Be2+ has such high charge density that it polarises anions strongly, creating shared electron density.
- Be shows a diagonal relationship with Al: both form amphoteric oxides, both are resistant to attack by concentrated nitric acid due to passivation, both form polymeric bridged halide structures.
Common Mistakes in s-Block Elements
Mistake 1: Writing that all alkali metals form normal oxides
Only Li forms a normal oxide. Na forms peroxide. K, Rb and Cs form superoxides. Getting this wrong in a NEET paper costs you a direct mark.
Mistake 2: Forgetting that Li3N forms directly with N2
Students know that N2 is generally unreactive. Li is the exception in Group 1 that directly forms a nitride with N2. This comes up as a PYQ regularly.
Mistake 3: Confusing the diagonal relationships
Li pairs diagonally with Mg. Be pairs diagonally with Al. B pairs diagonally with Si. Do not mix these up.
Mistake 4: Thinking Be(OH)2 is basic like other Group 2 hydroxides
Be(OH)2 is amphoteric. It dissolves in acid AND in base. Writing that it is only basic loses you the mark.
Quick Tip From Faculty: s-Block Anomalies
Think of Li and Be as “the outsiders of their groups.” They do not follow the group trends because they are unusually small for their family. Their small size creates high charge density, which drives all of their anomalous behaviour. When you see any question about anomalous behaviour in Groups 1 and 2, the answer almost always involves Li or Be. And when a question asks about diagonal relationships, it is always Li-Mg, Be-Al or B-Si. Knowing this pattern lets you answer s-Block anomaly questions in under 20 seconds.
NEET Dropper Year Plan 2026 — The Complete Month-by-Month Strategy to Go From Last Attempt to MBBS
p-Block Elements: The Most Feared Chapter Made Manageable
Let me say something directly before we begin. p-Block Elements is the longest chapter in Inorganic Chemistry. It covers Groups 13 through 18, spans two years of content, and includes dozens of compounds, reactions, properties and exceptions.
Students open it, feel overwhelmed by the volume, and either skip it or study it so superficially that they retain almost nothing by exam day.
Here is what those students are missing: p-Block is not difficult. It is large. And large content becomes completely manageable once you have a framework that organises it. I am going to give you that framework now, and then walk you through every group in the depth the exam actually requires.
The Framework That Reduces the Apparent Volume by 60 Percent
For every group in p-Block, there are exactly four things the exam tests:
- The general trend in oxidation states and where the inert pair effect kicks in
- The acidic or basic character of oxides and how it changes down the group
- One or two key compounds unique to this group with specific structures or properties
- The exceptions and anomalous behaviour of the first element in the group
If you systematically cover these four points for each of the six groups, you have covered 80 to 85 percent of all NEET and JEE questions from p-Block. The remaining 15 to 20 percent comes from knowing specific reactions and properties at a deeper level for the high-weightage groups (15, 16 and 17).
Let us go through each group with this framework.
Group 13 and 14: Diborane, CO2 vs SiO2 and the Inert Pair Effect
Group 13 (Boron Family: B, Al, Ga, In, Tl)
The key compound from Group 13 that appears in both NEET and JEE almost every year is Diborane (B2H6).
Why does BH3 not exist as a simple molecule? Because boron has only 3 valence electrons and forms 3 bonds, leaving an empty p orbital. This makes BH3 electron-deficient and highly unstable. It immediately dimerises to form B2H6.
The structure of B2H6 has two types of bonds. Four terminal B-H bonds are normal 2-centre 2-electron bonds. Two bridging B-H-B bonds are 3-centre 2-electron bonds, also called banana bonds. These bridging bonds involve only 2 electrons shared across 3 atoms, which is what makes diborane an electron-deficient compound.
The inert pair effect in Group 13: Thallium (Tl) predominantly shows the +1 oxidation state despite being in Group 13 where +3 is expected. The 6s2 electrons in Tl are held very tightly due to relativistic effects and do not participate in bonding. The +1 state (Tl+) is more stable than +3 (Tl3+). This inert pair effect gets stronger going down Groups 13, 14, 15 and 16, and the exam tests it for Tl, Pb, Bi and Po.
Group 14 (Carbon Family: C, Si, Ge, Sn, Pb)
The most important concept in Group 14 for both NEET and JEE is the comparison between CO2 and SiO2.
CO2 is a gas at room temperature. Its structure is a simple linear molecule (O=C=O) with two C=O double bonds. Carbon forms strong pi bonds because the 2p orbitals of C overlap very effectively.
SiO2 is a hard solid with a giant covalent network structure. Silicon cannot form strong pi bonds because its 3p orbitals are larger and more diffuse, so pi overlap is poor. Instead, each Si atom forms four sigma bonds to oxygen atoms, creating an extended three-dimensional network that runs throughout the crystal. This is why quartz and sand are hard solids, not gases.
This structural difference, carbon forming stable pi bonds and silicon not doing so, explains many other contrasts between C and Si. It explains why graphite exists for carbon but silicon has no graphite analogue. It explains why silicones are polymeric chains while simple carbon analogues are not. This concept appears across multiple question types in JEE and directly in NEET.
Pb predominantly shows +2 oxidation state due to the inert pair effect, while C and Si show +4.
Group 15: Oxoacids of Phosphorus and Oxides of Nitrogen
Group 15 (Nitrogen Family: N, P, As, Sb, Bi)
The concept that generates the most questions from Group 15, year after year, is the basicity of oxoacids of phosphorus. Students consistently get confused about this. Let me give you the rule that makes it impossible to get wrong again.
The one rule for basicity of phosphorus oxoacids:
Count only the P-OH groups. Each P-OH group provides exactly one ionisable proton. P-H bonds do not ionise because the P-H bond is not polar enough to release a proton.
- Hypophosphorous acid (H3PO2): Contains 1 P-OH group and 2 P-H bonds. Monobasic.
- Phosphorous acid (H3PO3): Contains 2 P-OH groups and 1 P-H bond. Dibasic.
- Phosphoric acid (H3PO4): Contains 3 P-OH groups and 0 P-H bonds. Tribasic.
This rule works for every phosphorus oxoacid the exam throws at you. Never count total H atoms. Count P-OH groups only.
Oxides of Nitrogen: Colours and Oxidation States
The colour of NO2 as a brown gas is directly tested in NEET. The equilibrium between brown NO2 and colourless N2O4 is a Le Chatelier’s principle question that appears in both NEET and JEE. At lower temperatures, the equilibrium shifts toward colourless N2O4 because the forward reaction is exothermic.
PCl5 Structure and the Bond Length Question
PCl5 has a trigonal bipyramidal structure with sp3d hybridisation. The three equatorial positions hold P-Cl bonds at 120 degrees to each other. The two axial positions hold P-Cl bonds at 90 degrees to the equatorial bonds.
Here is the subtlety the exam tests: the axial bonds in PCl5 are longer and weaker than the equatorial bonds. Why? The axial bond pairs experience repulsion from three equatorial bond pairs at 90 degrees, which is closer and more repulsive than the 120-degree separation between equatorial bonds. This extra repulsion weakens the axial bonds slightly. Students who say all five P-Cl bonds in PCl5 are equal get the comparison question wrong.
Group 16: Sulphur Chemistry and Allotropes
Group 16 (Chalcogens: O, S, Se, Te, Po)
The oxoacids of sulphur are tested regularly in both NEET and JEE. Here are the ones you must know with their oxidation states and key properties:
- H2SO3 (Sulphurous acid): S is in +4 oxidation state. Acts as a reducing agent.
- H2SO4 (Sulphuric acid): S is in +6 oxidation state. Strong diprotic acid. Concentrated H2SO4 is a dehydrating agent and a hot concentrated oxidising agent.
- H2S2O7 (Pyrosulphuric acid or Oleum): Formed by dissolving SO3 in concentrated H2SO4.
- H2S2O8 (Peroxodisulphuric acid): Contains an O-O peroxide linkage. Strong oxidising agent.
The key property of concentrated H2SO4 that NEET tests: it is a dehydrating agent. It removes water from sugar (C12H22O11) to leave a black carbon residue. It dehydrates HCOOH to give CO gas. These reactions appear as direct NEET questions.
Allotropes of Sulphur
Three allotropes come up in NEET papers:
- Rhombic sulphur (alpha-sulphur): Stable form below 96 degrees Celsius. Yellow crystals with eight-membered puckered ring structure (S8).
- Monoclinic sulphur (beta-sulphur): Stable form above 96 degrees Celsius. Needle-shaped yellow crystals, also S8 rings.
- Plastic sulphur: Formed by pouring molten sulphur into cold water. Amorphous, rubber-like, dark brown.
The transition temperature between rhombic and monoclinic sulphur is 96 degrees Celsius. This exact number appears as a direct NEET question. Above 96 degrees C, monoclinic is stable. Below 96 degrees C, rhombic is stable.
Group 17: Halogen Oxoacids and the F-F Bond Exception
Group 17 (Halogens: F, Cl, Br, I, At)
The F-F Bond Strength Exception
The bond dissociation enthalpy of X-X bonds follows this order: F2 less than Cl2 greater than Br2 greater than I2.
Students expect F2 to have the strongest bond because F is the smallest halogen and smaller atoms form shorter, stronger bonds. But F2 is actually weaker than Cl2. The reason is that F atoms are so small that when two F atoms bond, their lone pairs are very close to each other. The lone pair-lone pair repulsion between the two F atoms significantly weakens the F-F bond. Cl atoms are larger, the lone pairs are farther apart when bonded, and Cl-Cl has less lone pair repulsion and a stronger bond.
Oxoacids of Chlorine and Acidic Strength
This is one of the most reliably tested concepts in Group 17. The oxoacids of chlorine in increasing order of acidic strength are:
HOCl (Hypochlorous) less than HOClO (Chlorous) less than HOClO2 (Chloric) less than HOClO3 (Perchloric)
Acidic strength increases with increasing number of oxygen atoms bonded to chlorine. Each additional oxygen atom is strongly electronegative and pulls electron density away from the O-H bond. This weakens the O-H bond, making proton donation easier and the acid stronger.
The reason behind the trend is what the exam actually tests. Memorising the order without the reason leaves you unable to answer questions phrased in reverse or in a different format.
Bleaching Powder
Bleaching powder is a mixture of Ca(OCl)Cl, calcium hypochlorite and calcium chloride. It bleaches through the release of nascent oxygen:
CaOCl2 + CO2 gives CaCO3 + Cl2
Cl2 + H2O gives HCl + [O]
The nascent oxygen [O] is the actual bleaching agent. It oxidises the dye and destroys its colour. The bleaching is due to oxidation, not due to Cl2 directly. This distinction appears as a direct NEET question.
Group 18: Noble Gases and Xenon Compounds
Group 18 (Noble Gases: He, Ne, Ar, Kr, Xe, Rn)
Noble gases have completely filled valence shells, making them extremely stable under normal conditions. However, the heavier noble gases, Xe and Kr, can form compounds because their larger size and lower ionisation energy allow bonding with strongly electronegative atoms like F and O.
Xenon fluorides you must know:
- XeF2: sp3d hybridisation on Xe. 2 bond pairs and 3 lone pairs. Electron geometry is trigonal bipyramidal. Lone pairs occupy the equatorial positions (less repulsion there). Molecular shape is linear.
- XeF4: sp3d2 hybridisation on Xe. 4 bond pairs and 2 lone pairs. Electron geometry is octahedral. The two lone pairs occupy opposite axial positions to minimise lone pair-lone pair repulsion. Molecular shape is square planar.
- XeF6: sp3d3 hybridisation on Xe. 6 bond pairs and 1 lone pair. Distorted octahedral molecular shape.
- XeO3: sp3 hybridisation on Xe. 3 bond pairs and 1 lone pair. Pyramidal molecular shape.
The VSEPR rules you learnt in Chemical Bonding apply exactly here. Use the same framework: total electron pairs determines hybridisation, lone pairs determine how the molecular shape differs from the electron geometry.
The Trick to Never Forgetting p-Block Reactions
Here is the system I have used with thousands of students over 15 years. It takes one evening to set up and pays dividends throughout your preparation.
For each of the six p-Block groups, make a single A4 page with exactly these five sections:
- Important compounds with formulas and oxidation states
- Reactions with oxygen, water and halogens
- Acidic or basic character of oxides
- Anomalous behaviour of the first element
- The one concept in this group that appears in PYQs most often
You now have six pages covering all testable p-Block content. Review these six pages once every 10 days during preparation. By the time the exam arrives, you will have seen every concept 6 to 8 times. Each review takes 20 minutes. That is the most efficient revision system for p-Block that exists.
Common Mistakes Students Make in p-Block Elements
Mistake 1: Counting total H atoms to determine basicity of phosphorus oxoacids
Students count all 3 H atoms in H3PO3 and call it tribasic. It is dibasic because only 2 of the 3 H atoms are in P-OH groups. One H is directly bonded to P and does not ionise.
Mistake 2: Getting the F-F bond exception wrong
Students expect F2 to have the strongest bond in Group 17. The correct order is Cl2 greater than F2 due to lone pair-lone pair repulsion in the compact F2 molecule.
Mistake 3: Writing NO2 as colourless
NO2 is the brown gas. N2O4 is the colourless dimer. These are frequently swapped in answers.
Mistake 4: Getting XeF4 shape wrong as octahedral
XeF4 has octahedral electron geometry but square planar molecular shape. Students who do not account for lone pairs write “octahedral” and lose the mark.
Mistake 5: Saying all five bonds in PCl5 are equivalent
The axial bonds are longer and weaker than the equatorial bonds. Questions asking which bonds in PCl5 are longer appear in JEE papers specifically.
Mistake 6: Thinking bleaching powder bleaches by releasing Cl2
The bleaching agent is nascent oxygen, not Cl2. Questions asking why bleaching powder loses activity over time are answered by the fact that prolonged exposure to CO2 and moisture causes the hypochlorite to convert to carbonate, reducing the available nascent oxygen.
d and f Block Elements: How to Score Full Marks in 10 Days
Here is something that surprises students when I tell them. d and f Block Elements is one of the most predictable chapters in all of Inorganic Chemistry for NEET and JEE. The same concepts repeat year after year with remarkable consistency.
Once you know exactly which concepts get tested and understand the logic behind each one, this chapter becomes one of the fastest to prepare. I have had students go from zero to full marks in this chapter in 10 focused days. Let me show you exactly how.
Electronic Configurations: The Two Exceptions Every Student Must Know
The general electronic configuration of d-block elements is (n-1)d1-10 ns1-2. For the first row of transition metals (Sc to Zn), this means 3d and 4s orbitals are being filled.
Most elements follow this configuration predictably. Two elements do not, and these two exceptions appear in NEET and JEE papers every single year.
Chromium (Cr): Expected [Ar] 4s2 3d4. Actual [Ar] 4s1 3d5
One electron from 4s moves to 3d to give a half-filled 3d subshell. A half-filled subshell has extra stability because all five d orbitals each hold exactly one electron, maximising exchange energy and minimising repulsion. The system prefers this arrangement over the regular filling.
Copper (Cu): Expected [Ar] 4s2 3d9. Actual [Ar] 4s1 3d10
One electron from 4s moves to 3d to give a completely filled 3d subshell. A completely filled subshell is also extra stable. Cu sacrifices the regular configuration to achieve this stability.
These two exceptions, Cr and Cu, come up directly and indirectly in questions about electronic configurations, oxidation states, magnetic properties and compound formation. Know them so well that you can write them in your sleep.
Quick Tip From Faculty: Never Forget Cr and Cu
I teach my students to remember this with one sentence: “Chromium wants half, Copper wants full.” Cr wants a half-filled 3d5 subshell. Cu wants a completely filled 3d10 subshell. Both steal one electron from 4s to get what they want. That one sentence is all you need to remember both exceptions permanently.
Also note this for JEE: when forming ions, d-block elements lose electrons from 4s before losing from 3d. So Fe2+ has the configuration [Ar] 3d6, not [Ar] 4s2 3d4. Students who remove electrons from 3d first get the ionic configurations wrong and then get the magnetic moment calculation wrong downstream. Always remove 4s electrons first when forming ions.
All Six Properties of Transition Metals Explained With Reasons
Every property of transition metals has a root cause in their electronic structure. Learn each property with its reason. Exam questions test the reason, not just the property.
Property 1: Variable Oxidation States
Transition metals show multiple oxidation states because the energy difference between (n-1)d and ns orbitals is small. Electrons from both sets of orbitals can participate in bonding.
Manganese shows the widest range of any transition metal, from +1 to +7. Its common oxidation states are +2 (MnO, MnSO4), +4 (MnO2) and +7 (KMnO4). This wide range makes Mn a favourite for PYQs.
The stability of higher oxidation states: in the early transition metals (Sc, Ti, V, Cr, Mn), higher oxidation states are accessible and stable because there are many d electrons available for bonding. As you move toward the right (Fe, Co, Ni, Cu), lower oxidation states become more stable because the increasing nuclear charge holds the d electrons more tightly. Fe3+ is more stable than Fe5+. Cu+ and Cu2+ are common but Cu3+ is rare.
Property 2: Coloured Ions
Transition metal ions show colour because of d-d electronic transitions. When partially filled d orbitals are present, visible light is absorbed to promote a d electron from a lower energy d orbital to a higher energy d orbital. The complementary colour of the absorbed light is what we observe.
This only happens when d orbitals are partially filled. Two ions in the first transition series are colourless: Sc3+ with d0 configuration and Zn2+ with d10 configuration. With d0 there is no electron to promote. With d10 there is no empty orbital to accept a promoted electron. No d-d transition is possible in either case, so no visible light is absorbed and the ions appear colourless.
This is where marks hide in NEET. A question listing several transition metal ions and asking “which is colourless?” expects you to identify Sc3+ or Zn2+ based on their d0 and d10 configurations.
Ion colours you must know for NEET:
Property 3: Magnetic Properties
Magnetic behaviour depends entirely on unpaired electrons. Paramagnetic substances have one or more unpaired electrons and are attracted to magnetic fields. Diamagnetic substances have all electrons paired and are weakly repelled.
The magnetic moment formula:
Magnetic Moment (mu) = square root of n(n+2) Bohr Magnetons, where n is the number of unpaired electrons.
The ion with the highest magnetic moment in NEET questions is almost always Mn2+ or Fe3+, both with d5 high spin configurations and 5 unpaired electrons giving 5.92 BM.
For JEE: when calculating magnetic moment, write the full d-electron configuration of the ion first, then count unpaired electrons based on whether the ligand environment creates a high spin or low spin situation. Strong field ligands like CN- force electron pairing. Weak field ligands like F- and H2O allow electrons to remain unpaired.
Property 4: Catalytic Activity
Transition metals are excellent catalysts for two specific reasons. First, they have variable oxidation states, so they can accept and donate electrons within a reaction cycle by switching between oxidation states. Second, they can adsorb reactant molecules onto their surface because their partially filled d orbitals can interact with the bonding electrons of reactants, holding them close enough to react.
Catalysts every student must know by heart:
- Fe with Mo promoter: Haber process for NH3 synthesis
- V2O5: Contact process for SO3 synthesis
- Pt or Pd: Catalytic hydrogenation of unsaturated organic compounds
- Ni: Hydrogenation using Raney nickel
- MnO2: Decomposition of KClO3 to release O2
- Pt: Ostwald process (oxidation of NH3 to NO)
Property 5: Formation of Interstitial Compounds
Small atoms like hydrogen, carbon, nitrogen and boron can occupy the gaps (interstices) between metal atoms in the crystal lattice of transition metals. These interstitial compounds are harder than the pure metal, have higher melting points, are chemically inert and retain metallic conductivity.
Steel is the most familiar example: iron with carbon atoms in interstitial positions. The carbon atoms prevent the iron layers from sliding past each other, making steel much harder and stronger than pure iron.
Property 6: Alloy Formation
Transition metals form alloys readily because the atomic radii of adjacent transition metals are similar enough to allow one metal to substitute for another in the crystal lattice.
Alloys you must know:
- Brass: Cu and Zn
- Bronze: Cu and Sn
- Stainless steel: Fe, Cr and Ni
- Nichrome: Ni and Cr
KMnO4 and K2Cr2O7: The Two Compounds That Appear Every Year
These two compounds generate at least one question between them in every NEET paper and frequently in JEE Main too. Study them at this level of detail.
Potassium Permanganate (KMnO4)
Mn is in the +7 oxidation state in KMnO4. It is a powerful oxidising agent and its behaviour changes depending on the medium.
In acidic medium: MnO4- is reduced to Mn2+. The solution changes from purple to colourless. Change in oxidation state is 5.
MnO4- + 8H+ + 5e- gives Mn2+ + 4H2O
In neutral or faintly alkaline medium: MnO4- is reduced to MnO2. A brown precipitate forms.
MnO4- + 2H2O + 3e- gives MnO2 + 4OH-
In strongly alkaline medium: MnO4- is reduced to MnO42- (manganate ion). The solution turns green.
MnO4- + e- gives MnO42-
Preparation of KMnO4: MnO2 is fused with KOH in the presence of air or oxidising agent to form K2MnO4 (green, manganate). K2MnO4 is then electrolytically oxidised or treated with Cl2 to form KMnO4 (purple).
The acidic medium reaction is the one tested most often in NEET, specifically the decolourisation of KMnO4 by oxalic acid, Fe2+ ions, H2O2 and SO2. All four of these decolorise KMnO4 in acidic medium and all four have appeared as direct NEET questions.
Potassium Dichromate (K2Cr2O7)
Cr is in +6 oxidation state. Strong oxidising agent in acidic medium. Orange dichromate solution turns green as Cr3+ is formed.
Cr2O72- + 14H+ + 6e- gives 2Cr3+ + 7H2O
The orange to green colour change during oxidation reactions with K2Cr2O7 in acidic medium appears as a direct NEET question about colour changes in redox reactions.
Structure of the dichromate ion: Two CrO4 tetrahedra sharing one bridging oxygen. The Cr-O-Cr bond angle is approximately 126 degrees.
The pH-dependent interconversion between chromate and dichromate is tested directly in JEE:
-
Add acid: 2CrO42- (yellow, chromate) + 2H+ gives Cr2O72- (orange, dichromate) + H2O
-
Add base: Cr2O72- (orange) + 2OH- gives 2CrO42- (yellow) + H2O
This interconversion is reversible. Acidic conditions favour orange dichromate. Basic conditions favour yellow chromate.
Lanthanide Contraction and the f-Block Comparison
The lanthanides are the 14 elements from Ce to Lu, filling the 4f orbitals. The actinides are the 14 elements from Th to Lr, filling the 5f orbitals.
Lanthanide Contraction: What It Is and Why It Matters
As you move across the lanthanide series from La to Lu, atomic radius decreases steadily with each element. This is the lanthanide contraction. The reason is that 4f electrons, because of the diffuse shape of the f subshell, shield the outer electrons from the nuclear charge very poorly.
As each proton is added across the lanthanide series, nuclear charge increases but the poor shielding by 4f electrons means the outer electrons feel more of that increased nuclear pull. The electron cloud contracts steadily. The ionic radius of La3+ is 103 pm while that of Lu3+ is only 86.1 pm, a contraction of nearly 17 pm across 14 elements.
The consequences of lanthanide contraction that get tested in JEE:
First: The 4d and 5d transition metals in each group have nearly identical atomic radii, despite being one full period apart. Normally elements in the same group show increasing atomic radius going down. But the lanthanide contraction in between the 4d and 5d series counteracts the expected increase. Zr and Hf have almost the same atomic radius (160 pm and 159 pm respectively). This makes them extremely difficult to separate in ore processing, which has industrial importance.
Second: The 5d elements are generally denser, harder and have higher melting points than their 4d counterparts because their similar radii mean stronger metallic bonding with a higher nuclear charge.
Lanthanides vs Actinides: What NEET Tests
Common Mistakes in d and f Block Elements
Mistake 1: Writing Cu as [Ar] 4s2 3d9
The correct configuration of Cu is [Ar] 4s1 3d10. This is the most common d-block configuration error in NEET papers. Know the actual configurations of Cr and Cu perfectly.
Mistake 2: Removing electrons from 3d before 4s when forming ions
When Fe forms Fe2+, it loses two 4s electrons first. Configuration of Fe2+ is [Ar] 3d6. Students who remove 3d electrons first write [Ar] 4s2 3d4, which gives a completely wrong magnetic moment.
Mistake 3: Saying Sc3+ or Zn2+ is coloured
Both are colourless. d0 and d10 configurations cannot undergo d-d transitions. Any question about colourless d-block ions points to Sc3+ or Zn2+.
Mistake 4: Using the wrong medium for KMnO4 reactions
KMnO4 gives Mn2+ in acid, MnO2 in neutral and MnO42- in strong alkali. Mixing up the products across different media is a very common error.
Mistake 5: Confusing chromate colour with dichromate colour
Chromate (CrO42-) is yellow. Dichromate (Cr2O72-) is orange. Students frequently swap these. Acidic conditions give orange. Basic conditions give yellow.
Coordination Compounds: The Chapter That Separates Good Scores From Great Ones
Coordination Compounds is the chapter I always say has the best return on investment in all of Inorganic Chemistry. Three to four questions in NEET and two to three in JEE Main appear from this chapter almost every year.
The students who score full marks from this chapter are not the ones who read it most. They are the ones who understood the basic framework early and then practised applying it across PYQs. Let me give you that framework now.
Basic Terminology You Must Know Cold
Every concept in Coordination Compounds builds on these definitions. If any of these is unclear, the rest of the chapter will not settle properly.
Central Metal Atom or Ion: The metal at the centre of the complex that accepts electron pairs from ligands. It acts as a Lewis acid.
Ligand: A molecule or ion that donates a lone pair of electrons to the central metal. Ligands are Lewis bases. Examples: NH3, H2O, CN-, Cl-, F-, en (ethylenediamine), EDTA.
Coordination Number: The total number of coordinate bonds formed between the central metal and ligands. Not the same as the number of ligand molecules when multidentate ligands are involved. In [Co(en)3]3+, there are 3 ligands but the coordination number is 6 because each en forms 2 bonds.
Denticity: The number of donor atoms in a single ligand that bond to the metal.
- Monodentate: 1 donor atom (NH3, H2O, Cl-, CN-)
- Bidentate: 2 donor atoms (en or ethylenediamine, oxalate C2O42-)
- Hexadentate: 6 donor atoms (EDTA forms 6 bonds to metal)
Chelate: A complex in which a polydentate ligand uses two or more donor atoms to bond to the same metal, forming a ring. Chelates are more stable than comparable complexes with monodentate ligands. This extra stability is the chelate effect.
Inner Sphere vs Outer Sphere: Ligands inside the square bracket are directly bonded to the metal (inner sphere). Counter ions outside the bracket are held by electrostatic forces only (outer sphere). In [Co(NH3)5Cl]Cl2, the inner sphere contains Co3+, 5 NH3 and 1 Cl-. The outer sphere holds 2 Cl- ions. In solution, this gives 3 ions total: one [Co(NH3)5Cl]2+ and two Cl-.
The number of ions produced in solution determines the electrical conductance, depression of freezing point and osmotic pressure. These property-based questions appear in JEE Main regularly.
IUPAC Nomenclature in Six Steps
NEET and JEE name coordination compounds almost every year. Follow these six steps and you will name any complex correctly.
Step 1: Name the cation first, then the anion. Same convention as for ionic compounds.
Step 2: Within the complex ion, name ligands first in alphabetical order, then the central metal.
Step 3: Name anionic ligands with the suffix -o replacing the terminal -e:
- Cl- becomes chlorido
- Br- becomes bromido
- CN- becomes cyanido
- OH- becomes hydroxido
- O2- becomes oxido
- NO2- becomes nitrito-N (if bonded through N) or nitrito-O (if bonded through O)
Step 4: Neutral ligands keep their names with two classic exceptions everyone must know:
- H2O is called aqua
- NH3 is called ammine (two m’s, not one)
Step 5: Number prefixes for ligands:
- di, tri, tetra, penta, hexa for simple ligand names
- bis, tris, tetrakis, pentakis for complex ligand names (those that already contain a number, like ethylenediamine)
Step 6: Show the oxidation state of the central metal in Roman numerals in parentheses after the metal name. If the complex is an anion overall, the metal name takes the suffix -ate. Some metals use Latin names in anionic complexes: Fe becomes ferrate, Cu becomes cuprate, Au becomes aurate.
Practice these examples until they are automatic:
- [Co(NH3)6]Cl3: hexaamminecobalt(III) chloride
- [CoCl2(NH3)4]Cl: tetraamminedichloridocobalt(III) chloride
- K2[PtCl4]: potassium tetrachloridoplatinate(II)
- [Co(en)2Cl2]+: bis(ethylenediamine)dichloridocobalt(III) ion
- K3[Fe(CN)6]: potassium hexacyanidoferrate(III)
Quick Tip From Faculty: The Ammine Spelling Error That Costs Marks
In 15 years of correcting student answers, the most consistent single spelling error in Coordination Compounds is “amine” with one m instead of “ammine” with two m’s. Objective questions in NEET and JEE include options that differ only in this spelling. The reason for two m’s is to distinguish ammine (NH3 as a ligand in coordination chemistry) from amine (organic nitrogen compounds). Two m’s. Every time. Write it correctly once for every ten times you practise it until the double m is automatic.
All Six Types of Isomerism With Examples
Isomerism gives the most questions per unit of study time in this entire chapter. If you master all six types completely, you are securing at least 2 of the 3 or 4 marks this chapter contributes in NEET.
Structural Isomers (same molecular formula, different bonding arrangements)
Type 1: Ionisation Isomers
Same molecular formula but different ions inside and outside the coordination sphere.
[Co(NH3)5Br]SO4 and [Co(NH3)5SO4]Br are ionisation isomers.
The first compound releases Br- ions in solution. The second releases SO42- ions. Test them with AgNO3: the first gives a pale yellow AgBr precipitate, the second does not.
Type 2: Linkage Isomers
A ligand that can bond through two different donor atoms gives linkage isomers.
The nitrite ion NO2- is the classic example. It bonds through N to give a nitro complex: [Co(NH3)5(NO2)]2+ which is yellow. It bonds through O to give a nitrito complex: [Co(NH3)5(ONO)]2+ which is red-orange.
Thiocyanate SCN- bonds through S (thiocyanato-S) or through N (thiocyanato-N). These linkage isomers appear in JEE papers specifically.
Type 3: Coordination Isomers
Only possible in compounds where both the cation and anion are complex ions. Ligands redistribute between the two metal centres.
[Co(NH3)6][Cr(CN)6] and [Cr(NH3)6][Co(CN)6] are coordination isomers.
Type 4: Solvate or Hydrate Isomers
Different numbers of water molecules inside versus outside the coordination sphere.
The famous example is the three isomers of CrCl3.6H2O:
- [Cr(H2O)6]Cl3: violet, gives 3 Cl- ions in solution immediately
- [Cr(H2O)5Cl]Cl2.H2O: grey-green, gives 2 Cl- ions in solution
- [Cr(H2O)4Cl2]Cl.2H2O: dark green, gives 1 Cl- ion in solution
All three have the same molecular formula but different colours and different electrical conductance. NEET questions ask about this colour difference directly.
Stereoisomers (same bonds, different spatial arrangements)
Type 5: Geometrical Isomers (Cis-Trans)
In square planar complexes of type MA2B2: cis isomer has the two identical ligands adjacent to each other. Trans isomer has them on opposite sides.
The most tested example is the cisplatin and transplatin pair:
- cis-[PtCl2(NH3)2] is cisplatin: polar molecule (dipole moments of two Pt-Cl bonds point in the same direction and add up), biologically active anticancer drug.
- trans-[PtCl2(NH3)2] is transplatin: non-polar molecule (dipole moments cancel), not clinically effective.
The polarity difference and the anticancer activity of only the cis isomer appear as direct NEET questions.
In octahedral complexes of type MA4B2: cis isomer has the two B ligands at 90 degrees (adjacent positions). Trans isomer has the two B ligands at 180 degrees (opposite positions).
Type 6: Optical Isomers
Optical isomers are non-superimposable mirror images. They rotate plane-polarised light in opposite directions. A complex shows optical isomerism only when it has no plane of symmetry.
The key rule for JEE: the cis form of [M(en)2X2]n+ is optically active (no plane of symmetry). The trans form is not optically active (it has a plane of symmetry that makes it superimposable on its mirror image).
[M(en)3]3+ complexes are always optically active because the three bidentate ligands create a propeller-like arrangement with no plane of symmetry.
VBT vs CFT: What Each Exam Actually Tests
Valence Bond Theory (VBT)
VBT explains bonding in coordination compounds through hybridisation. The metal ion provides empty orbitals. Ligands donate electron pairs into those orbitals. The hybridisation determines the geometry.
The critical distinction for both NEET and JEE is inner orbital versus outer orbital complexes.
When strong field ligands like CN-, CO and NO2- are present, they force the 3d electrons to pair up, freeing inner 3d orbitals for hybridisation. The result is an inner orbital complex using d2sp3 hybridisation for octahedral geometry. These complexes tend to be diamagnetic or have fewer unpaired electrons.
When weak field ligands like F-, Cl-, Br-, H2O and OH- are present, 3d electrons remain unpaired and outer 4d orbitals are used in hybridisation. The result is an outer orbital complex using sp3d2 hybridisation for octahedral geometry. These complexes are paramagnetic with more unpaired electrons.
The textbook comparison that appears in NEET papers directly:
-
[Co(CN)6]3-: CN- is strong field. 3d electrons pair up. d2sp3 hybridisation. Diamagnetic.
-
[CoF6]3-: F- is weak field. 3d electrons remain unpaired. sp3d2 hybridisation. Paramagnetic.
Both contain Co3+ with d6 configuration, but the ligand field determines whether the complex is diamagnetic or paramagnetic.
Crystal Field Theory (CFT)
CFT explains colour and magnetic properties more accurately than VBT by considering the electrostatic effect of ligands on d orbital energies.
In an octahedral complex, ligands approach along the x, y and z axes. The d orbitals that point directly at the incoming ligands (dx2-y2 and dz2, called eg orbitals) are destabilised and pushed to higher energy. The d orbitals that point between the axes (dxy, dxz and dyz, called t2g orbitals) are stabilised and pushed to lower energy.
The energy gap between eg and t2g is the crystal field splitting energy, written as delta-o for octahedral complexes.
The spectrochemical series lists ligands from weakest to strongest field:
I- less than Br- less than S2- less than SCN- less than Cl- less than NO3- less than F- less than OH- less than ox2- less than H2O less than NCS- less than py less than NH3 less than en less than NO2- less than CN- less than CO
Weak field ligands (I-, Br-, Cl-) create small delta-o. Strong field ligands (CN-, CO) create large delta-o.
High spin versus low spin:
If delta-o is large (strong field ligand): electrons prefer to pair up in the lower t2g orbitals before occupying the higher eg orbitals. Result: low spin complex, fewer unpaired electrons, often diamagnetic or weakly paramagnetic.
If delta-o is small (weak field ligand): electrons follow Hund’s rule and occupy all available d orbitals singly before pairing. Result: high spin complex, more unpaired electrons, strongly paramagnetic.
This directly explains why [Fe(CN)6]4- is low spin (CN- is strong field, electrons pair in t2g, giving 0 unpaired electrons and diamagnetic behaviour) while [Fe(H2O)6]2+ is high spin (H2O is weak field, electrons spread across all d orbitals, giving 4 unpaired electrons and strongly paramagnetic behaviour). Both contain Fe2+ with d6 configuration. The ligand field is entirely responsible for the difference.
How CFT explains colour:
When an electron absorbs visible light and jumps from t2g to eg (a d-d transition), the wavelength absorbed corresponds to the energy gap delta-o. The complementary colour of the absorbed wavelength is transmitted and that is the colour we observe. Different ligands create different delta-o values, which means different wavelengths are absorbed and different colours are observed. This is why the same metal with different ligands shows different colours.
Common Mistakes Students Make in Coordination Compounds
Mistake 1: Confusing coordination number with number of ligands
Three bidentate ligands give a coordination number of 6, not 3. Always count bonds, not ligand molecules.
Mistake 2: Writing ammine with one m
This one error appears in objective questions designed specifically to catch it. Two m’s. Always. Ammine.
Mistake 3: Saying the trans form of [Co(en)2Cl2]+ is optically active
Only the cis form is optically active. The trans form has a plane of symmetry and is not optically active. This appears in NEET PYQs repeatedly.
Mistake 4: Confusing ionisation isomers with hydrate isomers
Both involve swapping ions inside and outside the coordination sphere. Hydrate isomers specifically involve water molecules being swapped in and out. Ionisation isomers involve non-water ions.
Mistake 5: Placing ligands in the wrong alphabetical order in IUPAC names
All ligands, regardless of charge, are listed alphabetically by their ligand name. The prefix (di, tri, bis, tris) is not considered in alphabetical ordering. Ammine comes before chlorido. Aqua comes before ammine. Alphabetical order by the base ligand name.
Mistake 6: Confusing d2sp3 with sp3d2
d2sp3 uses inner 3d orbitals and is formed with strong field ligands. sp3d2 uses outer 4d orbitals and is formed with weak field ligands. Both give octahedral geometry, but their magnetic properties are completely different.
Previous Year Question Analysis: Coordination Compounds
These question types have appeared in NEET from 2015 to 2025 with the highest frequency from this chapter. If you can answer all five confidently, you will not drop a mark from Coordination Compounds in NEET.
Type 1: Identify the type of isomerism between two given complexes
Appears every 2 years. The question gives two complexes with the same molecular formula and asks whether they are ionisation isomers, linkage isomers, geometrical isomers or optical isomers. Identify the key structural difference and match it to the isomerism type.
Type 2: Cisplatin structure, polarity and biological activity
Appeared in NEET 2016, 2019, 2021 and related questions in subsequent years. Know that cisplatin is the cis isomer of [PtCl2(NH3)2], that it is polar and that it is an anticancer drug. Know why the trans isomer is not effective.
Type 3: Which complex shows optical isomerism?
Appeared in NEET 2017, 2020, 2023. Answer is always the cis form of an octahedral complex with bidentate ligands like [Co(en)2Cl2]+, not the trans form.
Type 4: IUPAC naming
Appears almost every year in some form. Know the six steps and practise naming at least 20 different complexes before your exam.
Type 5: Magnetic moment from complex type
Gives a coordination compound and asks for magnetic moment or number of unpaired electrons. Write the electronic configuration of the metal ion. Determine high spin or low spin from the ligand in the spectrochemical series. Count unpaired electrons. Calculate magnetic moment using the formula.
How to Study Inorganic Chemistry: A Realistic 30-Day Strategy
The most common question I get from students is “Sir, I have one month left, can I still do well in Inorganic?” Yes. Absolutely yes. But only with this system.
The core principle of Inorganic Chemistry revision is this: your first read gives you 40 percent retention. Your third read gives you 80 percent. Your fifth read gives you 95 percent. Every strategy that works is built around getting you to 4 to 5 reads of the high-priority content before exam day.
Days 1 to 3: Chemical Bonding
Day 1: Read NCERT Chemical Bonding completely. Build your VSEPR table from scratch. Make a list of every exception you encounter.
Day 2: MOT section. Write out the filling sequences for N2 and O2 families separately. Calculate bond orders and magnetic properties of 10 diatomic species.
Day 3: Solve 30 PYQs from Chemical Bonding (NEET 2015 to 2025). Every wrong answer goes into your running exceptions list.
Days 4 to 6: Periodic Table and s-Block
Day 4: All six periodic trends. Write each trend with its mechanism, not just the direction. Then write every exception with its reason.
Day 5: s-Block complete NCERT. Focus specifically on anomalous behaviour of Li and Be and the three diagonal relationships.
Day 6: Solve 25 PYQs covering both chapters. Add wrong answers to exceptions list.
Days 7 to 13: p-Block Elements
Day 7: Group 13 and 14. Diborane structure. CO2 vs SiO2. Inert pair effect with examples from Tl and Pb.
Day 8: Group 15. Phosphorus oxoacid basicity rule applied to all three acids. Nitrogen oxides table with colours.
Day 9: Group 16. Sulphuric acid properties (dehydrating, oxidising, oleum). Sulphur allotropes with transition temperature of 96 degrees.
Day 10: Group 17. Oxoacids of chlorine with acidic strength order and mechanism. F-F bond exception with reason. Bleaching powder mechanism.
Day 11: Group 18. Xenon fluoride and oxide structures using VSEPR. XeF2, XeF4, XeF6 shapes cold.
Day 12: Make your six A4 summary pages for Groups 13 to 18 using the framework: compounds, reactions, acid-base character, anomalous behaviour of first element, top PYQ concept.
Day 13: Solve 50 PYQs across all p-Block groups. Build your second targeted exceptions list.
Days 14 to 17: d and f Block Elements
Day 14: NCERT complete read. Cr and Cu configurations absolutely confirmed. All six properties with reasons.
Day 15: Ion colours table. Magnetic moment calculations for 8 different d-block ions.
Day 16: KMnO4 and K2Cr2O7 in detail. All three media for KMnO4. pH-dependent interconversion for K2Cr2O7. Preparation of KMnO4.
Day 17: Lanthanide contraction, f-block comparison, all catalysts list. Solve 30 PYQs.
Days 18 to 24: Coordination Compounds
Day 18: All basic terminology until definitions are immediate. Denticity examples for mono, bi and hexadentate ligands.
Day 19: IUPAC nomenclature rules 1 to 6. Name 20 complexes in sequence without referring to rules.
Day 20: All six isomerism types with one example each. Cisplatin vs transplatin in full detail.
Day 21: VBT. Inner vs outer orbital for Co3+ complexes. High spin vs low spin with field strength.
Day 22: CFT. t2g and eg splitting. Spectrochemical series from memory. High spin vs low spin distinction for Fe2+ and Co3+ with different ligands.
Day 23: Solve 40 PYQs from Coordination Compounds. This is non-negotiable. This chapter rewards PYQ practice more than any other.
Day 24: Review every wrong answer from Days 18 to 23 carefully.
Days 25 to 27: Full Inorganic Revision Cycle
Day 25: Review your complete exceptions list. Every exception should be recalled with its reason, not just the statement.
Day 26: Re-read all six p-Block summary pages and Coordination Compounds notes.
Day 27: Full mock of Inorganic Chemistry only. Identify remaining gaps. Target them specifically.
Days 28 to 30: Final Consolidation
Day 28: Colour table, magnetic moment table, KMnO4 reactions in three media, K2Cr2O7 colour changes.
Day 29: Full Chemistry mock paper. Note every Inorganic question you answer wrong and revisit only those concepts.
Day 30: Light NCERT re-read of chapter headings and highlighted text. No new learning. Only reinforcement.
What Topper Students Do Differently in Inorganic Chemistry for NEET and JEE
The gap between a student scoring 120 in Chemistry and one scoring 170 is rarely about intelligence. After 15 years of watching thousands of students prepare, I can tell you with complete confidence that topper behaviour in Inorganic Chemistry is specific, repeatable and learnable by any student willing to adopt it.
Here is what toppers actually do differently, habit by habit.
Habit 1: They Read NCERT Line by Line, Not Paragraph by Paragraph
Average students skim NCERT looking for “important points.” Toppers read every single line because they know that NEET questions are written directly from NCERT text, sometimes almost word for word.
The difference is not the number of hours spent reading. It is the attention paid to each sentence. Toppers treat NCERT like a question paper, asking “how could this line become a question?” every time they read something new. When they encounter an exception, a reaction condition, or a specific property, they flag it immediately. They do not assume they will remember it from a single read.
The practical implication for you: read NCERT with a pencil or highlighter in hand. Mark every exception, every anomalous behaviour, every specific condition mentioned for a reaction. These are your future exam questions sitting right in front of you.
Habit 2: They Maintain a Living Exceptions List
This is the single habit I see most consistently in students who crack NEET with 160+ in Chemistry. They maintain what I call a living exceptions list.
From the first day they start Inorganic, every time they encounter a fact that breaks the general trend, they write it in one dedicated notebook with the exception and its reason side by side. Not just “Cl has more negative EGA than F” but “Cl has more negative EGA than F because F’s compact 2p orbital causes lone pair-lone pair repulsion when the incoming electron is added, which Cl’s larger 3p orbital avoids.”
By the time their exam arrives, this notebook has 40 to 60 carefully noted exceptions. They review this notebook once every 5 to 7 days during preparation. This means by exam day they have seen every exception 8 to 10 times. Their recall under pressure is automatic.
Average students rely on their memory from a single reading. Toppers build a system that makes forgetting nearly impossible.
Habit 3: They Do PYQs Chapter by Chapter Before Moving On
Most students complete a chapter and then immediately move to the next one. Toppers complete a chapter, immediately solve every NEET and JEE Main PYQ from that chapter going back 10 years, and only then move forward.
This habit does three things at once. It reveals exactly which concepts from that chapter actually get tested. It exposes the specific wording and phrasing the exam uses, which trains pattern recognition. And it shows the student where their understanding has gaps while the chapter is still fresh in their mind, not three weeks later.
If you solve 30 PYQs from Chemical Bonding immediately after reading it, you will discover that the exam tests shape-hybridisation mismatches, MOT-based magnetic properties and bond angle comparisons far more than it tests Lewis structures or ionic bonds. You will naturally focus your revision on what actually earns marks.
Habit 4: They Revise on a Strict 3-Read Cycle
Toppers understand that Inorganic Chemistry retention requires multiple exposures. They plan their schedule specifically to complete at least 3 reads of every chapter before the exam day, with the third read happening in the final week.
First read: Understanding. Building the framework. Noting exceptions.
Second read: Strengthening. Filling gaps. Adding to exceptions list.
Third read: Confirming. Everything should feel familiar. Any concept that does not feel automatic gets extra attention.
Average students do one full read and then “revise” by skimming their notes. That approach gives 40 to 50 percent retention. The 3-read cycle gives 85 to 90 percent retention. The difference shows directly in the exam score.
Habit 5: They Connect Concepts Across Chapters
The highest-scoring students in JEE specifically do something that average students never do: they actively look for connections between chapters.
The size-stability rule you learn in s-Block explains halide stability trends. The electronegativity trend you learn in Periodic Table explains bond polarity in Chemical Bonding and oxoacid strength in p-Block. The CFT you learn in Coordination Compounds explains the colours of transition metal ions you learn in d-Block.
When you study with connections, revising one concept reinforces three or four others simultaneously. This multiplies the efficiency of every revision session. It also prepares you for integrated questions in JEE Main that combine two or three chapter concepts in a single problem.
Habit 6: They Use Errors as Their Best Study Material
Every wrong answer in a PYQ or mock test is a gift if you treat it correctly. Toppers analyse wrong answers at three levels:
Level 1: What was the correct answer?
Level 2: Why was my answer wrong? (Conceptual gap, misread question, or careless error?)
Level 3: What do I need to understand more deeply so this exact error never happens again?
Average students check the answer key, note the correct answer and move on. This means they make the same mistake again in the actual exam.
Toppers write a one-line note for every wrong answer: “I chose X because I thought Y. The correct answer is Z because of [specific reason].” This note goes into their exceptions list. They are building a personalised exam guide from their own errors. By the day of their exam, their error log IS their most valuable revision tool.
Previous Year Question Pattern Analysis: Full Inorganic Chemistry for NEET and JEE
Understanding the question pattern is not optional. It is the difference between studying what the exam tests and studying what you assume the exam tests. These two things are not always the same.
Based on analysis of NEET papers from 2015 to 2025 and JEE Main papers from 2015 to 2025, here is the complete picture.
How Inorganic Chemistry Questions Have Changed Over the Years
From 2009 to 2013, both NEET and JEE Inorganic questions were primarily memory-based. State the colour of this compound. Write the formula of that product. Identify this element from its configuration.
From 2014 to 2017, the shift began. Application-based questions increased. Instead of “What is the colour of KMnO4?”, questions became “KMnO4 is used to oxidise oxalic acid in acidic medium. What colour change is observed and why?” The answer still requires the same knowledge but now requires you to apply it.
From 2018 onwards, two consistent trends emerged that are still visible in the most recent papers. First, exception-based questions increased dramatically. Nearly 40 percent of all Inorganic questions now involve a property or behaviour that does not follow the expected trend. Second, integrated questions that draw from two or more chapters in a single question became common in JEE Main.
The practical implication for 2026 preparation: pure memorisation is no longer sufficient. You need to understand every exception with its reason. You need to be able to apply periodic trends and bonding concepts to situations you may not have seen before.
Chapter-wise PYQ Frequency (NEET 2015 to 2025)
The Three Question Types That Appear Most in NEET Inorganic
Question Type 1: Exception Identifier
“Which of the following does NOT follow the expected trend?”
OR
“The ionisation enthalpy of which of the following is higher than expected?”
These questions test whether you know the exceptions with their reasons. They appear in Periodic Table, Chemical Bonding, p-Block and d-Block. Approximately 35 to 40 percent of all NEET Inorganic questions fall into this category. Your exceptions list is your direct preparation tool for these.
Question Type 2: Structure and Property Linker
“Which shape does XeF4 adopt and why?”
OR
“Why is cisplatin an anticancer drug but transplatin is not?”
These questions give a compound and ask you to connect its structure to a specific property. They require both knowledge and the ability to reason from structure to consequence. Approximately 25 to 30 percent of NEET Inorganic questions are of this type.
Question Type 3: Reaction Product or Colour Identifier
“What product is formed when KMnO4 reacts with FeSO4 in acidic medium?”
OR
“What colour does [Cu(NH3)4]2+ appear in aqueous solution?”
These test specific factual knowledge about well-defined reactions and properties. They are the most directly memorisable question type. Approximately 30 to 35 percent of NEET Inorganic questions fall here. These are the questions where your revision tables and summary pages pay off most directly.
What JEE Main Tests Differently From NEET
JEE Main Inorganic questions share most of the same content as NEET but have two important differences in pattern.
First, JEE Main uses integer-type and numerical answer questions for Inorganic Chemistry. These require precise knowledge, not elimination among four options. Magnetic moment calculations, coordination number identification, counting ions produced in solution and calculating oxidation states are all candidates for integer-type questions.
Second, JEE Main tests CFT and VBT at a greater depth than NEET. Questions asking you to calculate the number of unpaired electrons in a complex given the ligand field strength appear regularly in JEE Main but rarely in NEET. Knowing the spectrochemical series and being able to determine high spin versus low spin for any given metal-ligand combination is JEE-specific preparation that pays off directly.
Key Takeaways: Inorganic Chemistry for NEET and JEE 2026
Every student who reads through this guide and then asks “but what are the most important things to remember?” gets this list from me. These are the ten things that, if you know them deeply, will secure you the highest proportion of marks from Inorganic Chemistry for the time invested.
- Chemical Bonding is the highest weightage chapter. Hybridisation with the lone pair framework, VSEPR for shapes, and MOT for magnetic properties cover 90 percent of its questions. The molecular shape is never the same as electron geometry when lone pairs are present.
- The two IE exceptions that appear every year are Be greater than B and N greater than O. Both happen because of extra stable fully filled and half-filled subshells. Know them with reasons.
- F has lower electron gain enthalpy than Cl because F’s compact 2p orbital causes excessive repulsion when an incoming electron is added. This is one of the most tested exceptions in all of Inorganic Chemistry.
- In s-Block, Li and Be are the anomalous members of their groups. Li behaves like Mg. Be behaves like Al. These diagonal relationships have specific testable similarities that repeat in NEET every year.
- In p-Block, basicity of phosphorus oxoacids is determined only by the number of P-OH groups, not total H atoms. H3PO3 is dibasic, not tribasic.
- The electronic configurations of Cr ([Ar] 4s1 3d5) and Cu ([Ar] 4s1 3d10) are exceptions that every d-Block question directly or indirectly tests. Know these perfectly.
- Sc3+ (d0) and Zn2+ (d10) are colourless. Every other first-row transition metal ion with partial d filling shows colour through d-d transitions.
- KMnO4 gives Mn2+ (colourless) in acid, MnO2 (brown) in neutral and MnO42- (green) in strong alkali. KMnO4 starts purple in every medium.
- Coordination Compounds optical isomerism: only the cis form of [M(en)2X2] is optically active. The trans form has a plane of symmetry and is not optically active.
- Ammine is always spelled with two m’s. CN- ligand is cyanido. NH3 ligand is ammine. H2O ligand is aqua. These three ligand names appear in almost every IUPAC naming question.
Frequently Asked Questions: Inorganic Chemistry for NEET and JEE
Is Inorganic Chemistry easy to score in NEET?
Yes, Inorganic Chemistry is one of the most reliable scoring sections in NEET if prepared correctly. It contributes 10 to 13 questions carrying 40 to 52 marks. Because most questions are directly based on NCERT concepts, exceptions and reactions, a student who reads NCERT thoroughly multiple times and practises PYQs consistently can score 80 to 90 percent from this section. The challenge is not difficulty but volume and revision consistency.
How many times should I read NCERT for Inorganic Chemistry?
A minimum of three complete reads of NCERT is required for good retention in Inorganic Chemistry. The first read builds your understanding of the framework. The second read strengthens it and fills gaps. The third read, done in the final week before the exam, confirms everything and brings automatic recall. Students aiming for 160+ in Chemistry typically complete four to five reads with PYQ practice between each read.
Which is the most important chapter in Inorganic Chemistry for NEET?
Chemical Bonding and Coordination Compounds are tied for the most important chapters in Inorganic Chemistry for NEET. Both consistently contribute 3 to 4 questions per paper. Chemical Bonding has the highest marks density in Class 11 Inorganic. Coordination Compounds is the single chapter that most directly separates good Chemistry scores from exceptional ones in Class 12 Inorganic. Both chapters require conceptual understanding, not just memorisation.
How do I remember all the exceptions in Inorganic Chemistry?
The most effective system is a dedicated exceptions notebook maintained throughout your preparation. Every time you encounter an exception, write the exception and its specific reason on the same line. Review this notebook every 5 to 7 days. Do not just write “F has lower EGA than Cl.” Write “F has lower EGA than Cl because F’s compact 2p orbital causes electron-electron repulsion with the incoming electron.” The reason is what makes the exception stick in long-term memory and makes you recall it correctly under exam pressure.
What is the difference between how NEET and JEE Main test Coordination Compounds?
NEET tests Coordination Compounds primarily through IUPAC naming, identifying isomerism types, cisplatin properties and optical isomerism questions. These are all concept identification questions with four options. JEE Main tests the same concepts but adds CFT-based questions (high spin vs low spin, spectrochemical series application), integer-type questions on magnetic moments and coordination numbers, and deeper VBT questions comparing inner and outer orbital complexes. JEE Main also tests the number of ions a complex produces in solution more frequently than NEET.
Can I complete Inorganic Chemistry in one month for NEET?
Yes, one month is enough to cover Inorganic Chemistry for NEET if you follow a structured day-by-day plan. The critical requirement is that you complete Chemical Bonding and Coordination Compounds in the first two weeks since these two chapters give the most marks. Distribute p-Block across Days 7 to 13, d and f Block across Days 14 to 17, and use the remaining days for revision cycles and PYQ practice. The 30-day plan provided in this guide is specifically designed for students starting from scratch one month before their exam.
Why do toppers score so much higher in Inorganic Chemistry than average students?
The gap comes from three specific habits that toppers maintain. First, they read NCERT line by line rather than skimming for “important points,” which catches every exception and specific condition the exam tests. Second, they practise PYQs chapter by chapter immediately after studying each chapter, which reveals exactly what the exam actually tests versus what they assumed it would test. Third, they maintain a living exceptions list that they review every 5 to 7 days, ensuring that every anomalous behaviour in the syllabus is retained through multiple exposures rather than a single read.
Is NCERT enough for Inorganic Chemistry in JEE Main?
NCERT covers approximately 85 to 90 percent of what JEE Main tests in Inorganic Chemistry. For this reason, thorough NCERT preparation is the foundation. The additional 10 to 15 percent that NCERT alone does not fully cover includes deeper CFT applications (high spin vs low spin for complex calculation questions), integer-type magnetic moment problems and some complex isomerism identification questions. For these, practising JEE Main PYQs from the past 5 years after completing NCERT thoroughly is the most targeted additional preparation you can do.
Conclusion
If you have read this far, you now have something most NEET and JEE aspirants do not have: a complete, chapter-by-chapter, concept-by-concept system for Inorganic Chemistry that has been built from 15 years of classroom teaching and thousands of student experiences.
The framework is in your hands. The 30-day plan is ready for you to start today. The exceptions lists are built into every section of this guide for you to transfer directly into your notebook.
What you do next determines your score in NEET 2026 and JEE Main 2026. Here is the action I recommend: go back to Stage 2 of this guide right now and read the Chemical Bonding section with your NCERT open beside you. Note every point from this guide that you had missed in your NCERT reading. Start your exceptions notebook tonight.
Every day you wait is one fewer revision cycle before exam day.
Explore More Expert Chemistry Guides on EduAiTutors:
- Organic Chemistry for NEET and JEE 2026: Reaction Mechanisms Made Simple
- Physical Chemistry Formulas and Shortcuts: Complete Chapter-wise Guide
- NEET 2026 Chemistry Full Syllabus Strategy: How to Score 160+ in 60 Days
- Coordination Compounds Deep Dive: Complete Theory and PYQ Workbook
- p-Block Elements Visual Summary: All Groups 13 to 18 on Six Pages





